
Chemistry, 26.07.2019 15:30 CadenSkinner2003
A0.100 l solution of 0.300 m agno3 is combined with a 0.100 l solution of 1.00 m na3po4. calculate the concentration of ag and po43– at equilibrium after the precipitation of ag3po4 (ksp = 8.89 × 10–17).

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 04:00
14. many depressants reduce small muscle control, making it harder for a. you to steer b. your mind to consider complex problems c. the eye to scan, focus, or stay still d. the kidneys to filter alcohol out of the bloodstream
Answers: 3

Chemistry, 22.06.2019 04:40
Listen base your answer to the question on the information below.propane is a fuel that is sold in rigid, pressurized cylinders. most of the propane in a cylinder is liquid, with gas in the space above the liquid level. when propane is released from the cylinder, the propane leaves the cylinder as a gas. propane gas is used as a fuel by mixing it with oxygen in the air and igniting the mixture, as represented by the balanced equation below.c3h8(g) + 5o2(g) → 3co2(g) + 4h2o() + 2219.2 kja small amount of methanethiol, which has a distinct odor, is added to the propane to consumers detect a propane leak. in methanethiol, the odor is caused by the thiol functional group (–sh). methanethiol, ch3sh, has a structure that is very similar to the structure of methanol.what is the correct structural formula for a molecule of methanethiol
Answers: 3

Chemistry, 22.06.2019 07:10
Provide a stepwise curved arrow mechanism that fully explains the outcome of the reaction shown below. oh нао* heat он
Answers: 2

Chemistry, 22.06.2019 11:00
Ais a mountain created from eruptions of lava, ash, rocks, and hot gases.
Answers: 1
You know the right answer?
A0.100 l solution of 0.300 m agno3 is combined with a 0.100 l solution of 1.00 m na3po4. calculate t...
Questions




English, 06.12.2021 01:50



Mathematics, 06.12.2021 01:50

Mathematics, 06.12.2021 01:50

English, 06.12.2021 01:50

Mathematics, 06.12.2021 01:50


Spanish, 06.12.2021 01:50


English, 06.12.2021 01:50

Mathematics, 06.12.2021 01:50



Social Studies, 06.12.2021 01:50


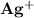 is 0.02998 M while the concentration of
is 0.02998 M while the concentration of 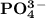 is 0.0999 M.
is 0.0999 M.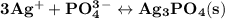
![\rm \bold{ Ksp = [ Ag^+]^3[ PO_4^3^- ]}](/tpl/images/0135/4274/43c55.png)
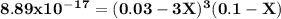
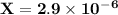
![\rm \bold{ [ Ag^+] = 0.02998 M}\\\\\\rm \bold{ [PO_4^3^-] = 0.0999 M }](/tpl/images/0135/4274/20a20.png)



