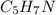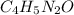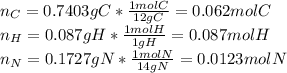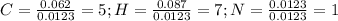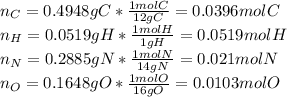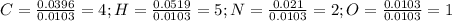Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 10:30
Asample of air with a volume of 2.20m3 at a pressure of 105 kpa and a temperature of 30c is cooled to 10c and the pressure is reduced to 75.0 kpa. what is the new volume? 6.9 1.34 2.56 43.0 2.88
Answers: 1

Chemistry, 22.06.2019 12:40
When 13.3 g koh is dissolved in 102.7 g of water in a coffee-cup calorimeter, the temperature rises from 21.4 °c to 31.53 °c. what is the enthalpy change per gram of koh (j/g) dissolved in the water? * take the density of water as 1.00 g/ml. * assume that the solution has a specific heat capacity of 4.18 j/g*k. enter to 1 decimal place. do not forget the appropriate sign /(+). canvas may auto-delete the (+) sign
Answers: 2

Chemistry, 23.06.2019 03:10
Which is true according to the law of conservation of energy
Answers: 1

Chemistry, 23.06.2019 05:00
Match the term to its description match term definition chemical energy a) internal energy caused by vibrations of atoms and molecules electrical energy b) electromagnetic energy that travels in waves radiant energy c) the movement of an electrical charge thermal energy d) potential energy stored in the bonds between atoms
Answers: 1
You know the right answer?
Calculate the empirical formula for each stimulant based on its elemental mass percent composition....
Questions


English, 22.01.2021 14:00

Mathematics, 22.01.2021 14:00


English, 22.01.2021 14:00

Mathematics, 22.01.2021 14:00


Mathematics, 22.01.2021 14:00


Chemistry, 22.01.2021 14:00

Mathematics, 22.01.2021 14:00


Arts, 22.01.2021 14:00

Mathematics, 22.01.2021 14:00



Biology, 22.01.2021 14:00

Mathematics, 22.01.2021 14:00

History, 22.01.2021 14:00