Chemistry, 25.07.2019 08:30 itcelmairani
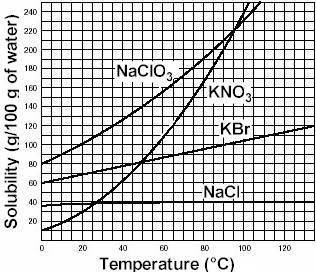
Asolution is a homogeneous mixture of one or more solutes dissolved in a solvent. a specific volume of solvent is only able to dissolve a limited amount of solute. as long as the solvent is able to dissolve more solute, the solution remains unsaturated. when the solvent can no longer dissolve additional solute, the solution is saturated. at this point, any additional solute will fall to the bottom of the container. the amount of solute that can be dissolved in a given amount of solvent at a specific temperature and pressure is defined as the solubility of the solute. consider the solubility curves of several salts in water. based on the information here, if 220 grams of the salt kbr are added to 100 ml of water at 100oc, we would label that solution as a) insoluble b) saturated c) supersaturated d) unsaturated

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 05:30
Which of the following two events occur to create a sea breeze? select all that apply. warm air rises on the ocean and moves toward the land to cool warm air rises on land and moves toward the ocean to cool cool air moves from the ocean to be warmed by the land cool air moves from the land to be warmed by the ocean
Answers: 3

Chemistry, 22.06.2019 11:40
Which of these expressions are correct variations of the combined gas law? p1v1t2 = p2v2t1 both
Answers: 2

Chemistry, 22.06.2019 12:30
Nebulae are enormous clouds in outer space. they are made mostly of hydrogen gas, helium gas, and dust. some nebulae glow brightly, while others do not. the stars that people see are huge, bright balls of glowing gas. they are made mostly of hydrogen and helium. which statement correctly describes other ways in which nebulae and stars are different? a. stars can form inside a nebula but a nebula can never be produced by any star. b. a star always has a higher density than a nebula. c. stars can never form inside a nebula but a nebula can be produced by any star. d. a nebula always has a higher density than a star.
Answers: 3
You know the right answer?
Asolution is a homogeneous mixture of one or more solutes dissolved in a solvent. a specific volume...
Questions






Mathematics, 14.12.2020 20:20


Mathematics, 14.12.2020 20:20


Mathematics, 14.12.2020 20:20

SAT, 14.12.2020 20:20



Mathematics, 14.12.2020 20:20


Chemistry, 14.12.2020 20:20