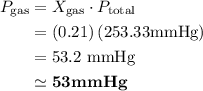Chemistry, 25.07.2019 07:00 ainhoagmz903
Atmospheric pressure at the summit of mount everest is about one third the pressure at sea level, which is 760 mm hg. if oxygen makes up 21% of the atmosphere by volume, the partial pressure of oxygen (po2) on mount everest is approximately atmospheric pressure at the summit of mount everest is about one third the pressure at sea level, which is 760 mm hg. if oxygen makes up 21% of the atmosphere by volume, the partial pressure of oxygen (po2) on mount everest is approximately 255 mm hg 53 mm hg 157 mm hg 760 mm hg

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 06:00
One of the few xenon compounds that form is cesium xenon heptafluoride (csxef7). how many moles of csxef7 can be produced from the reaction of 13.0 mol cesium fluoride with 12.5 mol xenon hexafluoride? csf(s) + xef6(s) csxef7(s)
Answers: 1

Chemistry, 22.06.2019 06:30
How many moles of carbon dioxide will form if 2.5 moles of c3h8 is burned
Answers: 1

Chemistry, 22.06.2019 07:30
According to the vsepr theory what is the shape of a molecule that has a central atom valence three other items with no lone pairs of electrons
Answers: 1

Chemistry, 22.06.2019 09:00
Identify the electromagnets with poles that are reversed from the electromagnet shown above
Answers: 3
You know the right answer?
Atmospheric pressure at the summit of mount everest is about one third the pressure at sea level, wh...
Questions


English, 25.10.2019 16:43

Health, 25.10.2019 16:43

Geography, 25.10.2019 16:43



History, 25.10.2019 16:43

English, 25.10.2019 16:43


Geography, 25.10.2019 16:43


History, 25.10.2019 16:43

Mathematics, 25.10.2019 16:43

Mathematics, 25.10.2019 16:43

Mathematics, 25.10.2019 16:43



Mathematics, 25.10.2019 16:43

Chemistry, 25.10.2019 16:43

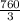 mmHg
mmHg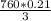 = 53.2 mmHg
= 53.2 mmHg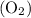 at Mount Everest is approximately 53 mmHg.
at Mount Everest is approximately 53 mmHg.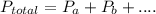 …… (1)
…… (1) is total pressure of the gas mixture.
is total pressure of the gas mixture.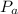 ,
, 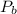 are the partial pressures of gas
are the partial pressures of gas  and
and 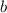 respectively.
respectively.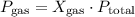 …… (2)
…… (2)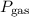 is the partial pressure of the gas.
is the partial pressure of the gas.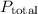 is the total pressure of the mixture.
is the total pressure of the mixture.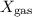 is the mole fraction of gas.
is the mole fraction of gas.
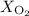 and 253.33 mmHg for
and 253.33 mmHg for 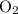 gas at Mount Everest.
gas at Mount Everest.