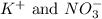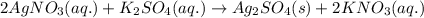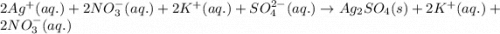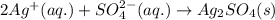Chemistry, 25.07.2019 03:30 Larkinlover703
What are the spectator ions in the solution after the precipitation of an insoluble sulfate compound from agno3(aq) and k2so4(aq)?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 07:30
Calculate the ratio of h+ ions to oh– ions at a ph = 7. find the concentration of h+ ions to oh– ions listed in table b of your student guide. then divide the h+ concentration by the oh– concentration. record this calculated ratio in table a of your student guide. compare your approximated and calculated ratios of h+ ions to oh– ions at a ph = 7. are they the same? why or why not? record your comparison in table a. what is the concentration of h+ ions at a ph = 7? mol/l what is the concentration of oh– ions at a ph = 7? mol/l what is the ratio of h+ ions to oh– ions at a ph = 7? : 1
Answers: 1

Chemistry, 22.06.2019 18:00
Which three statements represent the benefits of performing experiments using computer simulations?
Answers: 2

Chemistry, 22.06.2019 19:40
Scientists have developed an explanation of a phenomenon from several verified hypotheses. the explanation has been confirmed through numerous experimental tests.which option best describes this explanation? a. scientific lawb. research questionc. hypothesisd. scientific theory
Answers: 3

Chemistry, 23.06.2019 03:40
Write the overall equation for the reaction occurring in lithium battery?
Answers: 3
You know the right answer?
What are the spectator ions in the solution after the precipitation of an insoluble sulfate compound...
Questions


Physics, 09.07.2019 08:50


History, 09.07.2019 08:50

Mathematics, 09.07.2019 08:50

Advanced Placement (AP), 09.07.2019 08:50


Mathematics, 09.07.2019 08:50

Mathematics, 09.07.2019 08:50

English, 09.07.2019 08:50

History, 09.07.2019 08:50


History, 09.07.2019 08:50

English, 09.07.2019 08:50





Mathematics, 09.07.2019 08:50