
Chemistry, 24.07.2019 23:30 brodybb9350
The electrical conductivity of an aqueous solution depends on the concentration of which particles in the solution 1) molecules 2) electrons 3)atoms 4)ions

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 09:30
Melissa is interested in her family tree and how her family has changed over its many generations. melissa probably more closely resembles
Answers: 2

Chemistry, 22.06.2019 14:30
In water, a strong acid will break down into its component parts. a. completely b. partly c. never in water, a weak base will break down into its component parts. a. completely b. partly c. never
Answers: 2

Chemistry, 22.06.2019 20:00
In vapor-liquid equilibrium in a binary mixture, both components are generally present in both phases. how many degrees of freedom are there for such a system? the reaction between nitrogen and hydrogen to form ammonia occurs in the gas phase. how many degrees of freedom are there for this system? steam and coal react at high temperatures to form hydrogen, carbon monoxide, carbon dioxide, and methane. the following reactions have been suggested as being involved in the chemical transformation:
Answers: 3

Chemistry, 22.06.2019 21:00
The rate constant for the reaction below is 6.2 x 10−5 mol l−1 s −1. if the initial concentration of a is 0.0500 m, what is its concentration after 115 s?
Answers: 1
You know the right answer?
The electrical conductivity of an aqueous solution depends on the concentration of which particles i...
Questions




Biology, 23.03.2021 01:00

History, 23.03.2021 01:00


Computers and Technology, 23.03.2021 01:00



Arts, 23.03.2021 01:00


Mathematics, 23.03.2021 01:00



Mathematics, 23.03.2021 01:00

Mathematics, 23.03.2021 01:00


Mathematics, 23.03.2021 01:00


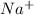 and
and 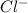 ions. Hence, flow of these ions will be responsible for electrical conductivity.
ions. Hence, flow of these ions will be responsible for electrical conductivity.

