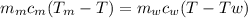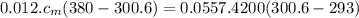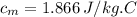Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 06:30
If 1.8 l of water is added to 2.5l of a 7.0 molarity koh solution, what is the molarity of the new solution
Answers: 1

Chemistry, 22.06.2019 07:30
According to the vsepr theory what is the shape of a molecule that has a central atom valence three other items with no lone pairs of electrons
Answers: 1

Chemistry, 23.06.2019 05:50
Which of the following is not a characteristic of s waves?
Answers: 1

Chemistry, 23.06.2019 08:10
An experiment is conducted to see if cats preferred skim milk or 2% milk. a cup of skim milkwas put out for 5 kittens and then measured how much the kittens drank over the course of aday. following a cup of 2% milk was purout for the skittens and then masured how much thekittens drank over the course of a day. the same kittens were used and the milk was served atthe same temperature. it was discovered that the cats liked the 2% milk more than the skimmilk. what is the dependent variable in this experiment?
Answers: 1
You know the right answer?
Apiece of metal of mass 12 g at 107 ◦c is placed in a calorimeter containing 55.7 g of water at 20◦c...
Questions

Social Studies, 01.12.2020 14:00

English, 01.12.2020 14:00


Mathematics, 01.12.2020 14:00

Mathematics, 01.12.2020 14:00


English, 01.12.2020 14:00

Mathematics, 01.12.2020 14:00

Biology, 01.12.2020 14:00

Mathematics, 01.12.2020 14:00


Chemistry, 01.12.2020 14:00


English, 01.12.2020 14:00




Mathematics, 01.12.2020 14:00

English, 01.12.2020 14:00

Mathematics, 01.12.2020 14:00