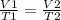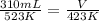Chemistry, 23.07.2019 09:00 Queenquestion9130
Asample of nitrogen gas has the temperature drop from 250.°c to 150.°c at constant pressure. what is the final volume if the initial volume is 310. ml?

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 16:30
Energy is released during which phase changes? check all that apply. boiling condensing depositing freezing melting subliming
Answers: 2

Chemistry, 21.06.2019 18:30
Which orbitals form a pi bond? a.the s orbital and three p orbitals b.the s orbital and two p orbitals c.overlapping p orbitals d.overlapping hybrid orbitals
Answers: 2

Chemistry, 22.06.2019 21:30
Athe top of a hill, an athlete on a skateboard has x joules of mechanical energy. how much mechanical energy will she have at the bottom of the hill? ignore the effects of friction.
Answers: 1

Chemistry, 23.06.2019 00:20
Steam reforming of methane ( ch4) produces "synthesis gas," a mixture of carbon monoxide gas and hydrogen gas, which is the starting point for many important industrial chemical syntheses. an industrial chemist studying this reaction fills a 1.5 l flask with 3.5 atm of methane gas and 1.3 atm of water vapor at 43.0°c. he then raises the temperature, and when the mixture has come to equilibrium measures the partial pressure of carbon monoxide gas to be 1 .0 atm. calculate the pressure equilibrium constant for the steam reforming of methane at the final temperature of the mixture. round your answer to 2 significant digits.
Answers: 1
You know the right answer?
Asample of nitrogen gas has the temperature drop from 250.°c to 150.°c at constant pressure. what is...
Questions


Mathematics, 10.05.2021 17:00





Mathematics, 10.05.2021 17:00



SAT, 10.05.2021 17:00

English, 10.05.2021 17:00


Mathematics, 10.05.2021 17:00


Mathematics, 10.05.2021 17:00


Mathematics, 10.05.2021 17:00

English, 10.05.2021 17:00

Mathematics, 10.05.2021 17:00

Mathematics, 10.05.2021 17:00