
Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 19:30
Determine the number o moles of ions/atoms/particle in the following: 2.50 miles of k2s (let me know how to do)
Answers: 1


Chemistry, 22.06.2019 17:10
Acalorimeter is to be calibrated: 51.203 g of water at 55.2 degree c is added to a calorimeter containing 49.783 g of water at 23.5c. after stirring and waiting for the system to equilibrate, the final temperature reached is 37.6 degree c. specific heat capacity of water (s = 4.18 j/g∙degree c). calculate the calorimeter constant. (smδt)warm water = -[(smδt)cold water + (calorimeterδtcold water)]
Answers: 2

Chemistry, 22.06.2019 22:30
Vi limitens. vastery test select the correct answer. which statement explains why large atoms are more reactive than small atoms? a. large atoms have valence electrons farther from the nucleus and lose them more readily. b. large atoms have greater ionization energy, which they can utilize during a reaction. c. large atoms have a greater number of electrons that they can lose during a reaction. d. large atoms have more energy levels, so they have more energy to pass on in a reaction. reset next
Answers: 3
You know the right answer?
Which salt is least soluble in water at 20c...
Questions


Chemistry, 05.11.2019 06:31

Mathematics, 05.11.2019 06:31



Social Studies, 05.11.2019 06:31

English, 05.11.2019 06:31



Geography, 05.11.2019 06:31


Mathematics, 05.11.2019 06:31

Mathematics, 05.11.2019 06:31



Spanish, 05.11.2019 06:31



History, 05.11.2019 06:31

Physics, 05.11.2019 06:31

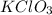 or potassium Chlorate has a low solubility in water hence among all other salts at 20 degree centigrade this one has the least chances of dissolving in it.
or potassium Chlorate has a low solubility in water hence among all other salts at 20 degree centigrade this one has the least chances of dissolving in it. 

