
Chemistry, 21.07.2019 23:30 dayjionneam
What volume of co2 gas at 645 torr and 800. k could be produced by the decomposition of 45.0 g of caco3? caco3(s) → cao(s) + co2(g) (r = 0.08206 l • atm/k • mol?

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 14:00
Diamond, graphite, and fullerenes share what property? a. they are all made of carbon (c) bonded to a metal. b. their shape. c. they are all made of carbon (c). d. they are all good conductors.
Answers: 1

Chemistry, 22.06.2019 00:00
What stress will shift the following equilibrium system to the left? n2(g) + 3h2(g) ⇌ 2nh3(g) adding more n2(g) adding more nh3(g) increasing the pressure of the system reducing the volume of the container
Answers: 1

Chemistry, 22.06.2019 02:30
When you perform this reaction, what could remain at the end of the reaction? check all that apply. excess reactant aqueous copper chloride excess reactant aluminum oxygen product solid copper carbon dioxide product aqueous aluminum chloride water
Answers: 2

Chemistry, 22.06.2019 06:30
If 1.8 l of water is added to 2.5l of a 7.0 m koh solution, what is the molarity of the new solution
Answers: 1
You know the right answer?
What volume of co2 gas at 645 torr and 800. k could be produced by the decomposition of 45.0 g of ca...
Questions

History, 26.03.2021 22:40

Mathematics, 26.03.2021 22:40


Mathematics, 26.03.2021 22:40

Mathematics, 26.03.2021 22:40




Advanced Placement (AP), 26.03.2021 22:40

Mathematics, 26.03.2021 22:40





Mathematics, 26.03.2021 22:40



Mathematics, 26.03.2021 22:40


Mathematics, 26.03.2021 22:40

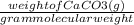
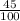 = 0.45 mol
= 0.45 mol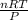 .
.


