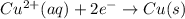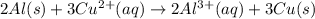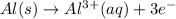Chemistry, 21.07.2019 18:30 theeflyguy5
Given the balanced ionic equation representing a reaction: 2al(s) + 3cu2+(aq) → 2al3+(aq) + 3cu(s)which half-reaction represents the reduction that occurs? al → al3+ +3eal3+ +3e → alcu→ cu2+ +2ecu2+ + 2e → cu

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 14:00
Anthracite is so hard and pure it is also referred to as a renewable resource metamorphic rock hot bituminous coal dirty fuel
Answers: 1

Chemistry, 22.06.2019 14:00
What is the ph of a solution that has a hydrogen ion concentration of 1.0 * 10 -9 m?
Answers: 2


Chemistry, 22.06.2019 20:30
Citric acid has a ph between 1 and 3. it is considered to be aa. weak acidb. weak basec. strong based. strong acid
Answers: 2
You know the right answer?
Given the balanced ionic equation representing a reaction: 2al(s) + 3cu2+(aq) → 2al3+(aq) + 3cu(s)w...
Questions


Mathematics, 08.04.2020 02:13













Mathematics, 08.04.2020 02:13

Social Studies, 08.04.2020 02:13