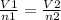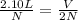Chemistry, 21.07.2019 09:00 itziabejar
If the number of moles of a gas initially contained in a 2.10 l vessel is doubled, what is the final volume of the gas in liters? (assume the pressure and temperature remain constant.) none of the above 8.40 4.20 1.05 6.30

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 05:00
Which position represents spring in the southern hemisphere? a) b) c) d)
Answers: 2

Chemistry, 22.06.2019 09:30
Which formula can be used to calculate the molar mass of hydrogen peroxide
Answers: 1

Chemistry, 22.06.2019 18:00
An object displaces 652 ml of water. the volume of the object is: 0.652 cm³ 6.52 cm³ 65.2 cm³ 652 cm³
Answers: 2

Chemistry, 22.06.2019 18:00
The fact that the total amount of energy in a system remains constant is a(n)
Answers: 1
You know the right answer?
If the number of moles of a gas initially contained in a 2.10 l vessel is doubled, what is the final...
Questions


Mathematics, 25.10.2021 21:40

Chemistry, 25.10.2021 21:40





Mathematics, 25.10.2021 21:40

Mathematics, 25.10.2021 21:40



Chemistry, 25.10.2021 21:40

History, 25.10.2021 21:40



Advanced Placement (AP), 25.10.2021 21:50


Social Studies, 25.10.2021 21:50