
Engineering, 25.03.2020 05:20 scbmaster351
3- A cylinder with 0.4kg of Nitrogen gas at 27ºC keeps under pressure by a frictionless piston. Weight of this piston will increase pressure to 0.35atm more than ambient pressure (which is 1atm) at 27ºC. Therefore, at the beginning Nitrogen is at 1.35atm and at equilibrium with surrounding. Consider these four processes which is happening in order:
A) System is reaching equilibrium after merging into a container of steam.
B) A variable force slowly pushing piston while temperature stay constant at 0ºC and Nitrogen compressed to half of its initial volume. At this point piston will stop by stoppers.
C) Taking the system out from steam container in order to reach equilibrium with surrounding.
D) Now remove stoppers until system reaches completely to equilibrium with surrounding.
Determine Q, W, ΔU, ΔH for the system. (consider Nitrogen as ideal gas)

Answers: 2
Another question on Engineering

Engineering, 03.07.2019 14:10
Explain the difference laminar and turbulent flow. explain it with the shear stress and the velocity profiles.
Answers: 1

Engineering, 03.07.2019 15:10
Apiston-cylinder with a volume of 0.25 m3 holds 1 kg of air (r 0.287 k/kgk) at a temperature of 100 c. heat transfer to the cylinder causes an isothermal expansion of the piston until the volume triples. how much heat is added to the piston-cylinder?
Answers: 3

Engineering, 04.07.2019 12:10
On a average work day more than work place firs are reorted
Answers: 1

Engineering, 04.07.2019 18:10
Steel is coated with a thin layer of ceramic to protect against corrosion. what do you expect to happen to the coating when the temperature of the steel is increased significantly? explain.
Answers: 1
You know the right answer?
3- A cylinder with 0.4kg of Nitrogen gas at 27ºC keeps under pressure by a frictionless piston. Weig...
Questions

Biology, 03.12.2021 05:00


Spanish, 03.12.2021 05:00





Computers and Technology, 03.12.2021 05:00

Mathematics, 03.12.2021 05:00





Mathematics, 03.12.2021 05:00

English, 03.12.2021 05:00


Computers and Technology, 03.12.2021 05:00

Computers and Technology, 03.12.2021 05:00

Social Studies, 03.12.2021 05:00

Medicine, 03.12.2021 05:00

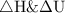 are zero because they are sate functions and in a cyclic process the change in state functions are zero,
are zero because they are sate functions and in a cyclic process the change in state functions are zero, 
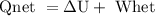 with appropriate sign convention , since
with appropriate sign convention , since 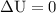 \
\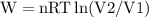
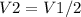
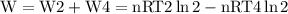
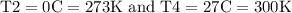
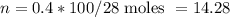
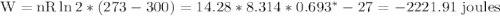 , so this amount of work has to be done on the system , since Q= W , Q = -2221.91 Joules of heat is to be transfered from the system.
, so this amount of work has to be done on the system , since Q= W , Q = -2221.91 Joules of heat is to be transfered from the system.

