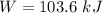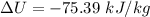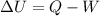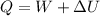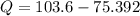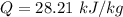Engineering, 06.04.2020 23:33 meadowsoares7
Nitrogen (N2) contained in a piston–cylinder arrangement, initially at 10 bar and 405 K, undergoes an expansion to a final temperature of 300 K, during which the pressure–volume relationship is pV1.3 = constant. Assuming the ideal gas model for the N2, determine the heat transfer in kJ/kg.

Answers: 3
Another question on Engineering

Engineering, 03.07.2019 14:10
Explain the difference laminar and turbulent flow. explain it with the shear stress and the velocity profiles.
Answers: 1

Engineering, 03.07.2019 14:10
When at a point two solid phase changes to one solid phase on cooling then it is known as a) eutectoid point b) eutectic point c) peritectic point d) peritectoid point
Answers: 3

Engineering, 03.07.2019 14:10
Amass of 1.5 kg of air at 120 kpa and 24°c is contained in a gas-tight, frictionless piston-cylinder device. the air is now compressed to a final pressure of 720 kpa. during the process, heat is transferred from the air such that the temperature inside the cylinder remains constant. calculate the boundary work input during this process.
Answers: 2

Engineering, 03.07.2019 14:10
Amass of m 1.5 kg of steam is contained in a closed rigid container. initially the pressure and temperature of the steam are: p 1.5 mpa and t 240°c (superheated state), respectively. then the temperature drops to t2= 100°c as the result of heat transfer to the surroundings. determine: a) quality of the steam at the end of the process, b) heat transfer with the surroundings. for: p1.5 mpa and t 240°c: enthalpy of superheated vapour is 2900 kj/kg, specific volume of superheated vapour is 0. 1483 m/kg, while for t 100°c: enthalpy of saturated liquid water is 419kj/kg, specific volume of saturated liquid water is 0.001043m/kg, enthalpy of saturated vapour is 2676 kj/kg, specific volume of saturated vapour is 1.672 m/kg and pressure is 0.1 mpa.
Answers: 3
You know the right answer?
Nitrogen (N2) contained in a piston–cylinder arrangement, initially at 10 bar and 405 K, undergoes a...
Questions

Health, 22.07.2019 23:50

History, 22.07.2019 23:50





History, 22.07.2019 23:50


History, 22.07.2019 23:50

Mathematics, 22.07.2019 23:50

Mathematics, 22.07.2019 23:50

Mathematics, 22.07.2019 23:50

Mathematics, 22.07.2019 23:50

Biology, 22.07.2019 23:50




Physics, 22.07.2019 23:50

Mathematics, 22.07.2019 23:50

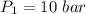
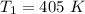
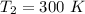
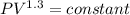
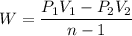
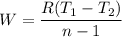
![W=\dfrac{0.296(405-300)}{1.3-1}\quad [R_{N_2}=\frac{8.314}{28}]](/tpl/images/0585/1869/e6619.png)