Engineering, 21.04.2020 16:30 camp591
A relatively nonvolatile hydrocarbon oil contains 4.0 mol % propane and is being stripped by direct superheated steam in a stripping tray tower to reduce the propane content to 0.2%. The temperature is held constant at 422 K by internal heating in the tower at 2.026 × 105 Pa pressure. A total of 11.42 kg mol of direct steam is used for 300 kg mol of total entering liquid. The vapor–liquid equilibria can be represented by y = 25x, where y is mole fraction propane in the steam and x is mole fraction propane in the oil. Steam can be considered as an inert gas and will not condense. Plot the operating and equilibrium lines and determine the number of theoretical trays needed.

Answers: 1
Another question on Engineering

Engineering, 04.07.2019 16:10
An electrical motor raises a 50kg load at a construct velencity .calculate the power of the motor, if it takes 40sec to raise the load through a height of 24m(take g =9.8n/g)
Answers: 2

Engineering, 04.07.2019 18:10
Air is to be cooled in the evaporator section of a refrigerator by passing it over a bank of 0.8-cm-outer-diameter and 0.4-m-long tubes inside which the refrigerant is evaporating at -20°c. air approaches the tube bank in the normal direction at 0°c and 1 atm with a mean velocity of 4 m/s. the tubes are arranged in-line with longitudinal and transverse pitches of sl- st 1.5 cm. there are 30 rows in the flow direction with 15 tubes in each row. determine (a) the refrigeration capacity of this system and (b) pressure drop across the tube bank. evaluate the air properties at an assumed mean temperature of -5°c and 1 atm. is this a good assumption?
Answers: 1

Engineering, 04.07.2019 18:10
Carbon dioxide gas expands isotherm a turbine from 1 mpa, 500 k at 200 kpa. assuming the ideal gas model and neglecting the kinetic and potential energies, determine the change in entropy, heat transfer and work for each kilogram of co2.
Answers: 2

Engineering, 04.07.2019 18:10
A-mn has a cubic structure with a0 0.8931 nm and a density of 7.47 g/cm3. b-mn has a different cubic structure, with a0 0.6326 nm and a density of 7.26 g/cm3. the atomic weight of manganese is 54.938 g/mol and the atomic radius is 0.112 nm. determine the percent volume change that would occur if a-mn transforms to b-mn.
Answers: 2
You know the right answer?
A relatively nonvolatile hydrocarbon oil contains 4.0 mol % propane and is being stripped by direct...
Questions


History, 15.01.2020 19:31




Geography, 15.01.2020 19:31

Physics, 15.01.2020 19:31




Physics, 15.01.2020 19:31


Mathematics, 15.01.2020 19:31





History, 15.01.2020 19:31

Spanish, 15.01.2020 19:31

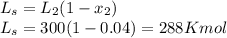
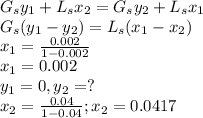
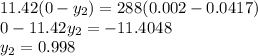
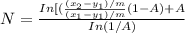
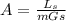
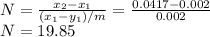
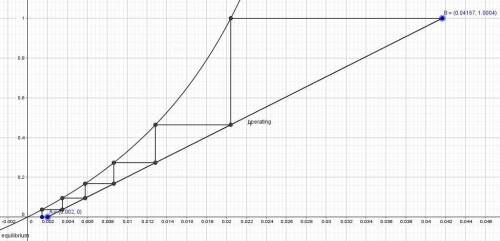
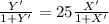 ......... 1
......... 1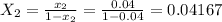
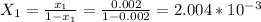
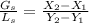 .............. a
.............. a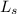 = 300(1 - 0.04) = 288 kmol pure oil
= 300(1 - 0.04) = 288 kmol pure oil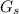 = 11.42 kmol
= 11.42 kmol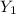 = 0 , solvent free steam
= 0 , solvent free steam