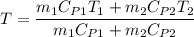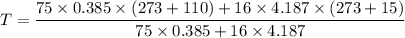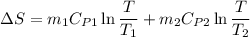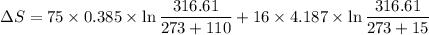Answers: 1
Another question on Physics

Physics, 21.06.2019 17:20
The specific heat of silver is 0.057 calories/gram°c. if 10.0 grams of silver were heated and the temperature of the sample changed by 20.0°c, how many calories of heat energy were absorbed by the sample?
Answers: 1

Physics, 22.06.2019 04:10
You will be galileo perform the experiment to determine if objects with different mass fall at the same, or different, rates in the air and in a vacuum. (refer to the walk-through video to locate the online lab within the online textbook).
Answers: 2

Physics, 22.06.2019 07:00
Examine the equation. 23490th→23088ra+42he what kind of barrier would you need to block the radioactive particles from this reaction? a.a piece of paper b.a sheet of aluminum foil c. a two-inch block of lead d. a solid concrete block
Answers: 1

Physics, 22.06.2019 13:50
9.98 kg of r-134a at 300 kpa fills a rigid container whose volume is 14 l. determine the temperature and total enthalpy in the container. the container is now heated until the pressure is 600 kpa. determine the temperature and total enthalpy when the heating is completed. use data from the steam tables.
Answers: 1
You know the right answer?
A75-kg copper block initially at 110°c is dropped into an insulated tank that contains 160 l of wate...
Questions

English, 02.12.2020 21:50

English, 02.12.2020 21:50







Business, 02.12.2020 21:50

Mathematics, 02.12.2020 21:50


English, 02.12.2020 21:50

Biology, 02.12.2020 21:50



English, 02.12.2020 21:50