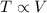Physics, 18.02.2020 05:36 marcaveli294
A mixture of gaseous reactants is put into a cylinder, where a chemical reaction turns them into gaseous products. The cylinder has a piston that moves in or out, as necessary, to keep constant pressure on the mixture of 1 atm. The cylinder is also submerged in a large insulated water bath. The temperature of the water bath is monitored, and it is determined from this data that 133.0 kJ of heat flows into the system during the reaction. The position of the piston is also monitored, and it is determined from the data that the piston does 241.0 kJ of work on the system during the reaction.
(1) Does the temperature of the water bath go up or down?
(2) Does the piston move in or out?
(3) Does heat flow into or out of the gaseous mixture?
(4) How much heat flows?

Answers: 2
Another question on Physics


Physics, 22.06.2019 19:20
The dipole moment of the water molecule (h2o) is 6.17x10^-30 c.m. consider a water molecule located at the origin whose dipole moment p points in the +x-direction. a chlorine ion ( of charge-1.60x10^-19c , is located at x=3.00x10^-9m . assume that is much larger than the separation d between the charges in the dipole, so that the approximate expression for the electric field along the dipole axis can be used. a) find the magnitude of the electric force that the water molecule exerts on the chlorine ion. b) what is the direction of the electric force. -x-direction or +x-direction c) is this force attractive or repulsive?
Answers: 1

Physics, 23.06.2019 10:30
Up of elements with the same number of valence electrons. vertical column in the periodic table of elements such as alkali metals or halogens. a horizontal row of elements in the periodic table. this is a characteristic of the elements in the periodic table that shows a pattern. it may increase or decrease across or down the table. periodic trend these are the highly reactive elements located in group 1 of the periodic table. these elements have one electron in their outer energy level which makes them highly reactive with water and halogens. these are the reactive elements located in group 2 of the periodic table. these elements have two electrons in their outer energy level which makes them reactive with water and halogens. alkaline earth metals these are the group 3 or d-block elements. these dense metals with high boiling points can have different oxidation states and all are solid at room temperature with the exception of mercury. transition metals this is the highly reactive family of elements with 7 valence electrons. this is an element with full valence shell, very unreactive. this is a group of elements with few valence electrons that conducts heat and electricity. one of a class of elements having properties intermediate to metals and nonmetals. this is a type of element that has many valence electrons, not a conductor.
Answers: 1

Physics, 23.06.2019 10:30
Suppose your surface body temperature averaged 90 degrees f. how much radiant energy in w/m^2 would be emitted from your body?
Answers: 1
You know the right answer?
A mixture of gaseous reactants is put into a cylinder, where a chemical reaction turns them into gas...
Questions



English, 29.06.2019 14:00

Computers and Technology, 29.06.2019 14:00

English, 29.06.2019 14:00

History, 29.06.2019 14:00


Mathematics, 29.06.2019 14:00

Mathematics, 29.06.2019 14:00






Biology, 29.06.2019 14:00

Chemistry, 29.06.2019 14:00


Mathematics, 29.06.2019 14:00

English, 29.06.2019 14:00

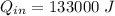 work done on the system,
work done on the system, 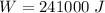 pressure on the system,
pressure on the system, 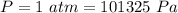
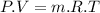
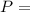 absolute pressure of the gas
absolute pressure of the gas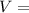 volume of the gas
volume of the gas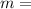 mass of the gas
mass of the gas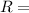 universal gas constant
universal gas constant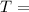 absolute temperature of the gas
absolute temperature of the gas