
Physics, 20.02.2020 08:16 madisontrosclair2
A cylinder contains oxygen gas at a pressure of 2.00 atm. The volume is 4.00 L, and the temperature is 300 K. Assume that the oxygen may be treated as an ideal gas. The oxygen is carried through the following processes:
(1) Heated at constant pressure from the initial state (state 1) to state 2, which has 450 K
(2) Cooled at constant volume to 250 K (state 3).
(3) Compressed at constant temperature to a volume of 4.00 L (state 4).
(4) Heated at constant volume to 300 K, which takes the system back to state 1.
A) Calculate Q and W for each of the four processes.
B) Calculate the net work done by the oxygen.
C) What is the effiency of this device as a heat engine? How does this effiiency compare with that of a Carnot-cycle engine operating between the same minimum temperatures of 250K and 450K?

Answers: 2
Another question on Physics

Physics, 21.06.2019 14:50
Ingrid wrote the hypothesis below. if the temperature of a liquid increases, the density of the liquid decreases because the particles move farther apart. what are the variables in her hypothesis?
Answers: 3

Physics, 22.06.2019 07:30
Write the function getkthdigit(n, k) that takes a possibly-negative int n and a non-negative int k, and returns the kth digit of n, starting from 0, counting from the right
Answers: 3

Physics, 22.06.2019 11:40
Imagine that you have two balloons (or, better yet, actually inflate two balloons, if possible). create static electricity around one of the balloons by rubbing it against your hair or your sweater and then bring that balloon close to the other balloon, which has not been charged. try this with at least one other object—and for variety in the discussion, avoid using an object already described by your classmates. then, for your initial post to the discussion, answer the following questions: what happened with the two balloons?
Answers: 3

Physics, 22.06.2019 13:40
Which of the following is not a transverse wave? a) soundb) lightc) radiod) all of thesee) none of these
Answers: 1
You know the right answer?
A cylinder contains oxygen gas at a pressure of 2.00 atm. The volume is 4.00 L, and the temperature...
Questions

Mathematics, 26.06.2019 23:30

Mathematics, 26.06.2019 23:30


Mathematics, 26.06.2019 23:30

History, 26.06.2019 23:30

Health, 26.06.2019 23:30

Social Studies, 26.06.2019 23:30

Advanced Placement (AP), 26.06.2019 23:30

Mathematics, 26.06.2019 23:30



History, 26.06.2019 23:30


Biology, 26.06.2019 23:30

Mathematics, 26.06.2019 23:30

Mathematics, 26.06.2019 23:30


Mathematics, 26.06.2019 23:30


Biology, 26.06.2019 23:30

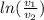 per unit mass of the gas
per unit mass of the gas

