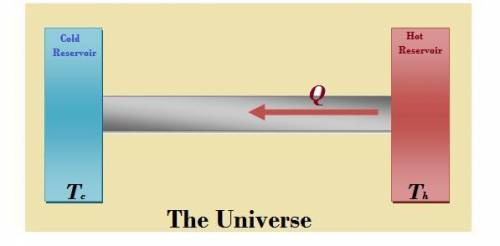
Physics, 21.02.2020 22:23 milkshakegrande101
A metal rod is used to conduct heat between two reservoirs at temperatures Th and Tc, respectively. When an amount of heat Q flows through the rod from the hot to the cold reservoir, what is the net entropy change of the rod, the hot reservoir, the cold reservoir, and the universe? (Use any variable or symbol stated above as necessary.)

Answers: 3
Another question on Physics

Physics, 21.06.2019 22:00
Aroll in a paper machine is powered by a separately excited dc machine. operating point 1 (under load): the roll typically operates at 764 rpm and must provide 301 nm of torque to oppose the load of the paper. the armature voltage is 230v for this operating point operating point 2 (no load): when the load is removed the speed of the roll increases to 900 rpm (the field characteristics are unchanged) la ea dev m (a) calculate the maximum torque (units of nm) that the motor can supply with this applied voltage (b) calculate the value of the armature resistance, ra (in ohms) (c) at operating point 1 (loaded condition), the field current, i, and the total input power (armature field), are measured to be to be 10 a and 28,600 w respectively. use this information to calculate the value of the field resistance, rf (in ohms). (d) (d) at operating point 1 (loaded condition), calculate the motor efficiency. express your answer as a percent to at least 2 significant digits. (d) if the field voltage is reduced by 1/2, calculate the new stall torque (in nm) of the motor (f). if the field voltage is reduced by 1/2, calculate the new no-load speed (in rad/sec)
Answers: 1

Physics, 22.06.2019 05:30
The volume of a gas is halved during an adiabatic compression that increases the pressure by a factor of 2.5. what is the specific heat ratio? show the math steps .
Answers: 3

Physics, 22.06.2019 11:50
Select all that applywhat are some basic resources a family is expected to provide for children? educationclothesspending
Answers: 2

Physics, 22.06.2019 18:50
An insulated thermos contains 148 g of water at 72.7 ˚c. you put in a 11.7 g ice cube at 0.00 ˚c to form a system of ice + original water. the specific heat of liquid water is 4190 j/kg•k; and the heat of fusion of water is 333 kj/kg. what is the net entropy change of the system from then until the system reaches the final (equilibrium) temperature?
Answers: 2
You know the right answer?
A metal rod is used to conduct heat between two reservoirs at temperatures Th and Tc, respectively....
Questions

Biology, 26.03.2020 01:30



Mathematics, 26.03.2020 01:31











English, 26.03.2020 01:32





Mathematics, 26.03.2020 01:33