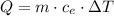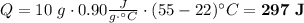Physics, 22.01.2020 18:31 KyiahDenise
How many joules of heat are needed to raise the temperature of 10.0 g of aluminum from 22 degrees celcius to 55 degrees celcius, if the specific heat of aluminum is 0.90 joules?

Answers: 1
Another question on Physics


Physics, 22.06.2019 13:00
The substances that are necessary for producing of certain hormones and that store and transport vitamins
Answers: 1

Physics, 22.06.2019 14:50
Nitrogen (n2) undergoes an internally reversible process from 6 bar, 247°c during which pν1.2 = constant. the initial volume is 0.1 m3 and the work for the process is 121.14 kj. assuming ideal gas behavior, and neglecting kinetic and potential energy effects, determine heat transfer, in kj, and the entropy change, in kj/s. show the process on a t-s diagram.
Answers: 2

You know the right answer?
How many joules of heat are needed to raise the temperature of 10.0 g of aluminum from 22 degrees ce...
Questions


History, 20.01.2021 21:50

Mathematics, 20.01.2021 21:50



Mathematics, 20.01.2021 21:50

Mathematics, 20.01.2021 21:50

Mathematics, 20.01.2021 21:50


English, 20.01.2021 21:50

English, 20.01.2021 21:50

Mathematics, 20.01.2021 21:50

Mathematics, 20.01.2021 21:50

Chemistry, 20.01.2021 21:50

Arts, 20.01.2021 21:50


Mathematics, 20.01.2021 21:50


French, 20.01.2021 21:50