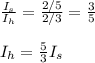Answers: 2
Another question on Physics

Physics, 22.06.2019 00:10
The energy released by a chemical reaction can be measured using a calorimeter. when barium hydroxide octahydrate crystals are reacted with dry ammonium chloride inside of a coffee cup calorimeter, the temperature of the 18.00 g of water in the calorimeter decreases from 30.0°c to 8.0°c. the equation for calculating energy absorbed or released by a reaction is: where q is the energy released or absorbed, m is the mass of water in the calorimeter, cp is the specific heat of water, and δt is the observed temperature change. if the specific heat of liquid water is 4.19 j/g·°c, how much energy was absorbed by the reaction?
Answers: 3

Physics, 22.06.2019 11:00
The dot diagram represents the motion of the motorcycle as it moves from left to right
Answers: 3


Physics, 22.06.2019 14:00
Explain why you think this diagram shows what happened to the carbon in the biodome.
Answers: 2
You know the right answer?
Two spheres have the same radius and equal masses. One is made of solid aluminum, and the other is m...
Questions



Biology, 31.08.2019 23:30

History, 31.08.2019 23:30


Biology, 31.08.2019 23:30



Social Studies, 31.08.2019 23:30

Physics, 31.08.2019 23:30

English, 31.08.2019 23:30





Mathematics, 31.08.2019 23:30




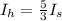
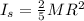 (1)
(1)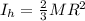 (2)
(2)