
Physics, 12.12.2020 16:30 cocodemain
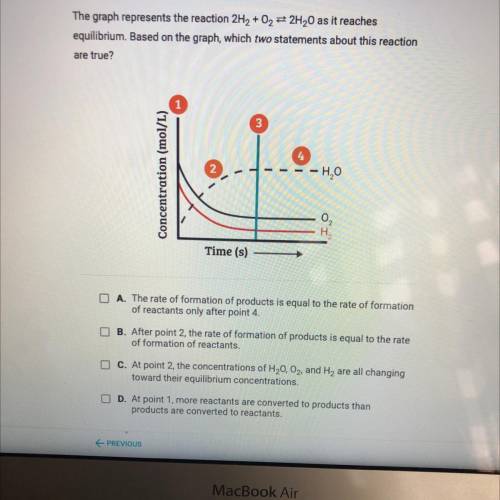
The graph represents the reaction 2H2 + 02 32H20 as it reaches
equilibrium. Based on the graph, which two statements about this reaction
are true?
Concentration (mol/L)
le
-H₂O
Time (s)
A. The rate of formation of products is equal to the rate of formation
of reactants only after point 4
B. After point 2, the rate of formation of products is equal to the rate
of formation of reactants.
C. At point 2, the concentrations of H20, O2, and H2 are all changing
toward their equilibrium concentrations.
D. At point 1. more reactants are converted to products than
products are converted to reactants.

Answers: 3
Another question on Physics

Physics, 22.06.2019 14:40
According to valence bond theory, which orbitals overlap in the formation of the bond in hf according to valence bond theory, which orbitals overlap in the formation of the bond in hf 2s on h and 2p on f 1s on h and 2s on f 1s on h and 1p on f 1s on h and 2p on f 1s on h and 3p on f
Answers: 3

Physics, 22.06.2019 18:30
Against his financial advisor's advice, frank has decided to invest his money in some risky stocks because he once made quite a bit of money in the stock market. his decision illustrates a. the representativeness heuristic b. overconfidence c. the availability heuristic d. confirmation bias select the best answer from the choices provided
Answers: 3


Physics, 23.06.2019 04:31
You have 80 g of 60oc water and 80 g or 10oc water. what is the final temperature when the two are mixed? explain.
Answers: 2
You know the right answer?
The graph represents the reaction 2H2 + 02 32H20 as it reaches
equilibrium. Based on the graph, whi...
Questions

History, 08.02.2021 21:10


Mathematics, 08.02.2021 21:10

Social Studies, 08.02.2021 21:10

Mathematics, 08.02.2021 21:10

Mathematics, 08.02.2021 21:10

Mathematics, 08.02.2021 21:10





Mathematics, 08.02.2021 21:10


Mathematics, 08.02.2021 21:10

Chemistry, 08.02.2021 21:10


Physics, 08.02.2021 21:10





