
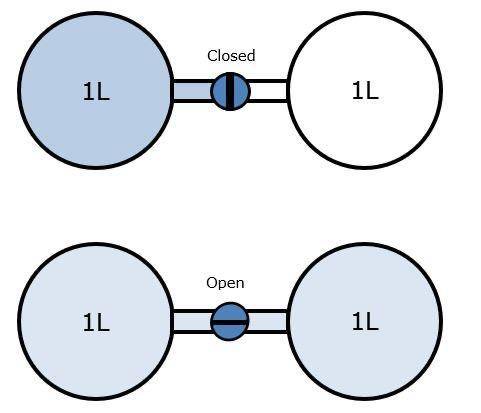
Two glass bulbs each having a volume of exactly 1L are connected by a valve. The left bulb contains a sample of nitrogen gas at a pressure of 101.325 kPa (kiloPascal), the right bulb has been emptied of gas and the pressure in the right bulb is 0 kPa.
(Use photo)
Which statement provides the BEST explanation for what happens when the valve between the bulbs is opened?
A
The volume available for the gas will double. Doubling the volume will cause the pressure and temperature to decrease.
B
The volume available for the gas will double. Doubling the volume will increases the amount of gas to keep the pressure constant.
C
The gas will expand into the empty bulb. Expansion of the gas will increase the gas pressure and decrease the gas temperature.
D
The gas will expand into the empty bulb. Expansion of the gas will increase the gas pressure and increase the gas temperature.

Answers: 3
Another question on Physics

Physics, 22.06.2019 11:00
Alarge box of mass m is pulled across a horizontal frictionless surface by a horizontal rope with tension t. a small box of mass m sits on top of the large box. the coefficients of static and
Answers: 1


Physics, 22.06.2019 21:00
What is the efficiency of an engine that does 80 j of work and exhausts 320 j of heat while taking in 400 j of heat ? a. 10% b. 20% c. 80% d. 25%
Answers: 1

Physics, 23.06.2019 01:00
First answer marked as brainiest which is the correct equation describing the first law of thermodynamics? a) when the internal energy of a system changes, it equals the amount of heat added plus the work done. b) when work is done by the piston, it is equal to the heat added plus the change in internal energy of the system. c) when heat is added to the system, it is equal to the amount of work done by the piston plus the change in internal energy of the system.
Answers: 2
You know the right answer?
Two glass bulbs each having a volume of exactly 1L are connected by a valve. The left bulb contains...
Questions

Chemistry, 03.09.2020 20:01

History, 03.09.2020 20:01

Mathematics, 03.09.2020 20:01


History, 03.09.2020 20:01

Chemistry, 03.09.2020 20:01



History, 03.09.2020 20:01

History, 03.09.2020 20:01


History, 03.09.2020 20:01

Engineering, 03.09.2020 20:01

Mathematics, 03.09.2020 20:01




Mathematics, 03.09.2020 20:01




