
Chemistry, 12.07.2019 05:30 myparentsrock17
Achemist adds 220.0ml of a 0.194 m potassium permanganate kmno4 solution to a reaction flask. calculate the mass in grams of potassium permanganate the chemist has added to the flask

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 15:00
What is the most important factor in determining climates.
Answers: 1

Chemistry, 22.06.2019 15:30
Each of the following reactions is allowed to come to equilibrium and then the volume is changed as indicated. predict the effect (shift right, shift left, or no effect) of the indicated volume change. drag the appropriate items to their respective bins.co(g) + h2o(g) < => co2(g) + h2(g) (volume is decreased) pcl3(g) + cl2(g) < => pcl5(g) (volume is increased) caco3(s)< => cao(s) + co2(g) (volume is increased)
Answers: 1

Chemistry, 22.06.2019 18:20
Which reason best explains why metals are malleable? a)because they have delocalized electrons b)because they have localized electrons c)because they have ionic bonds d)because they have rigid bonds
Answers: 2

Chemistry, 22.06.2019 21:00
The rate constant for the reaction below is 6.2 x 10−5 mol l−1 s −1. if the initial concentration of a is 0.0500 m, what is its concentration after 115 s?
Answers: 1
You know the right answer?
Achemist adds 220.0ml of a 0.194 m potassium permanganate kmno4 solution to a reaction flask. calcul...
Questions





Computers and Technology, 12.08.2020 20:01

Arts, 12.08.2020 20:01



Geography, 12.08.2020 20:01



English, 12.08.2020 20:01








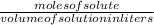 -(1)
-(1)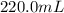 (given)
(given)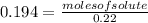
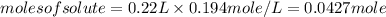
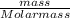 -(2)
-(2)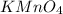 =
= 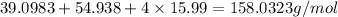
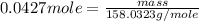
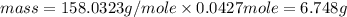
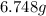 .
.

