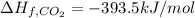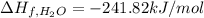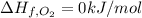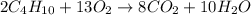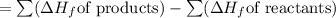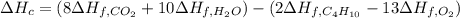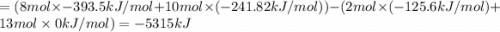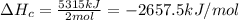Butane (c4 h10(g), hf = –125.6 kj/mol) reacts with oxygen to produce carbon dioxide (co2 , hf = –393.5 kj/mol ) and water (h2 o, hf = –241.82 kj/mol) according to the equation below. what is the enthalpy of combustion (per mole) of c4h10 (g)? use . –2,657.5 kj/mol –5315.0 kj/mol –509.7 kj/mol –254.8 kj/mol

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 04:20
Neils bohr believed that electrons orbited the nucleus in different energy levels, based on strong support from
Answers: 1

Chemistry, 22.06.2019 07:10
Remember to use the proper number of significant figures and leading zeros in all calculations.gelatin has a density of 1.27 g/cm³. if you have a blob of gelatin dessert that fills a 2.0 liter bottle, what is its mass? 2540 g2500 g3.9 x 10-43.937x 10-4
Answers: 3


Chemistry, 22.06.2019 14:00
How many absorptions would you expect to observe in the 13c nmr spectra of the following molecules? a) 3-chloropentane b) cis-4-methyl-2-pentene
Answers: 2
You know the right answer?
Butane (c4 h10(g), hf = –125.6 kj/mol) reacts with oxygen to produce carbon dioxide (co2 , hf = –393...
Questions