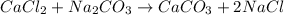cacl2 + na2co3 

Chemistry, 20.01.2020 04:31 rubixkube86
What are the products in the following double-displacement reaction?
cacl2 + na2co3 
a.
caco3 + 2nacl
b.
na2 + caco3 + cl2
c.
cao3 + 2nacl + c
d.
cao3 + na2ccl2
e.
cao2 + 2nacl(co)

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 11:50
Calculate the molarity of each of the following solutions. part a) 0.12 mol of lino3 in 5.5 l of solution part b) 60.7 g c2h6o in 2.48 l of solution part c) 14.2 mg ki in 100 ml of solution
Answers: 2

Chemistry, 22.06.2019 19:30
How might this scientific phenomena be explained? a paper clip floats on water.
Answers: 1

Chemistry, 22.06.2019 22:30
Calculate the concentration of all species in a 0.165 m solution of h2co3.
Answers: 1

Chemistry, 22.06.2019 23:00
The data below were determined for the reaction shown below. s2o82– + 3i – (aq) → 2so42– + i3– expt. # [s2o82–] (m) [i –] (m) initial rate 1 0.038 0.060 1.4 × 10 – 5 m/s 2 0.076 0.060 2.8 × 10 – 5 m/s 3 0.076 0.030 1.4 × 10 – 5 m/s the rate law for this reaction must be:
Answers: 1
You know the right answer?
What are the products in the following double-displacement reaction?
cacl2 + na2co3 
cacl2 + na2co3 
Questions











History, 17.08.2021 03:40

Mathematics, 17.08.2021 03:40



Mathematics, 17.08.2021 03:40


Mathematics, 17.08.2021 03:40


English, 17.08.2021 03:40

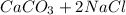
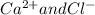 ) and (
) and (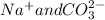 ) are two different ionic pairs. Reaction between these two pairs resulting exchange of ionic species between them. Hence
) are two different ionic pairs. Reaction between these two pairs resulting exchange of ionic species between them. Hence 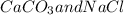 are formed as products of the given reaction.
are formed as products of the given reaction.