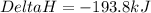Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 19:20
The equation picture below shows which type of nuclear reaction u 235 + n x e 134 + sr 100 + 2n
Answers: 1

Chemistry, 22.06.2019 19:50
When the mercury level in a barometer decreases that atmospheric pressure has
Answers: 3


Chemistry, 23.06.2019 00:30
Which of the following best describes technology a. something created for only scientists to use b.the method of thinking that scientists use. c.the application of engineering to create useful products. c. a scientific idea
Answers: 1
You know the right answer?
What is the heat of reaction when sulfur dioxide reacts with oxygen to form sulfur trioxide? 2so2(g...
Questions



Biology, 25.02.2020 00:02

Mathematics, 25.02.2020 00:02

Social Studies, 25.02.2020 00:02

Mathematics, 25.02.2020 00:02


Mathematics, 25.02.2020 00:02


Mathematics, 25.02.2020 00:02

Mathematics, 25.02.2020 00:02

Mathematics, 25.02.2020 00:02

Biology, 25.02.2020 00:02



Health, 25.02.2020 00:02




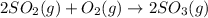
![\Delta H=\sum [n\times \Delta H_f(product)]-\sum [n\times \Delta H_f(reactant)]](/tpl/images/0273/9627/76c37.png)
![\Delta H=[(n_{SO_3}\times \Delta H_{SO_3})]-[(n_{O_2}\times \Delta H_{O_2})+(n_{SO_2}\times \Delta H_{SO_2})]](/tpl/images/0273/9627/23304.png)
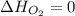 (as heat of formation of substances in their standard state is zero
(as heat of formation of substances in their standard state is zero![\Delta H=[(2\times -395.7)]-[(1\times 0)+(2\times -298.8)]](/tpl/images/0273/9627/57adc.png)