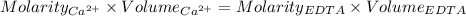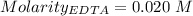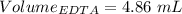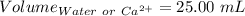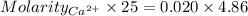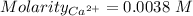Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 19:00
How does a catalyst increase the speed of a reaction? a. the catalyst eliminates the activated complex stage, allowing products to form immediately. b. the catalyst lowers the energy level of the reactants, making it easier for them to react. c. the catalyst makes it easier for the activated complex to form, lowering the activation energy. d. the catalyst raises the energy level of the products, making the reaction finish sooner. reset next
Answers: 1

Chemistry, 22.06.2019 21:30
How many liters of 3.0 m naoh solution will react with 0.60 liters of 4.0 m h2so4? h2so4 + naoh → na2so4 + h2o 1.2 l 1.6 l 2.4 l 2.8 l
Answers: 3

Chemistry, 22.06.2019 23:20
In medium-sized stars such as the sun, nuclear fusion almost always means the fusing of nuclei to form , but larger stars can produce elements as heavy as
Answers: 2

Chemistry, 23.06.2019 00:30
Five different substances are given to you to be dissolved in water. which substances are most likely to undergo dissolution in water? check all that apply. view available hint(s) check all that apply. sodium fluoride, naf octane, c8h18 propanol, ch3ch2ch2oh potassium iodide, ki benzene, c6h6
Answers: 1
You know the right answer?
Astudent titrates 25.00 ml. of tap water using 4.86 ml of 0.020 m edta. calculate the hardness of th...
Questions

Mathematics, 12.03.2021 01:20

Mathematics, 12.03.2021 01:20

Mathematics, 12.03.2021 01:20





English, 12.03.2021 01:20

Mathematics, 12.03.2021 01:20


Mathematics, 12.03.2021 01:20




Spanish, 12.03.2021 01:20



History, 12.03.2021 01:20


Mathematics, 12.03.2021 01:20

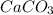 in water.
in water.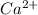 = Moles of EDTA
= Moles of EDTA