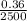Answers: 3
Another question on Chemistry

Chemistry, 23.06.2019 08:40
Which statement is true according to the kinetic theory? a. molecules of different gases with the same mass and temperature always have the same average density. b. molecules of different gases with the same mass and temperature always have the same average volume. c. molecules of different gases with the same mass and temperature always have the same pressure. d. molecules of different gases with the same mass and temperature always have the same molecular mass. e. molecules of different gases with the same mass and temperature always have the same average kinetic energy.
Answers: 1

Chemistry, 23.06.2019 12:30
What would happen to a weak base dissociation equilibrium if more products we added
Answers: 1


Chemistry, 23.06.2019 14:00
Which of the following represents the balanced reduction half-reaction from the redox reaction? (2 points) pb + pd(no3)2 yields pb(no3)2 + pd pb yields pd2+ + e- pd2+ + 2e- yields pd pb + e- yields pb 2 pd2+ + 4 e- yields 2 pd
Answers: 1
You know the right answer?
Sealed 2500 flask is full to capacity with 0.36 g of a substance. determine density of the substance...
Questions

Mathematics, 11.03.2020 22:25


Computers and Technology, 11.03.2020 22:25









Mathematics, 11.03.2020 22:26

English, 11.03.2020 22:26



English, 11.03.2020 22:26


Mathematics, 11.03.2020 22:26


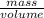 =
=