Chemistry, 24.09.2019 19:00 janwesley3
If the half-life of 37rb is 4.7x101 years, how long would it take for 0.5 grams of a 2 gram sample to radioactively decay?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 01:00
Consider three unlabeled bottles, each contain small pieces of one of the following metals. - magnesium - sodium - silver the following reagents are used for identifying the metals. - pure water - a solution of 1.0 molar hcl - a solution of concentrated hno3 (a) which metal can be easily identified because it is much softer than the other two? describe a chemical test that distinguishes this metal from the other two, using only one of the reagents above. write a balanced chemical equation for the reaction that occurs. (b) one of the other two metals reacts readily with the hcl solution. identify the metal and write the balanced chemical equation for the reaction that occurs when this metal is added to the hcl solution. use the table of standard reduction potentials (attached) to account for the fact that this metal reacts with hcl while the other does not. (c) the one remaining metal reacts with the concentrated hno3 solution. write a balanced chemical equation for the reaction that occurs. (d) the solution obtained in (c) is diluted and a few drops of 1 m hcl is added. describe what would be observed. write a balanced chemical equation for the reaction that occurs.
Answers: 2


Chemistry, 23.06.2019 05:00
In 1901, thomas edison invented the nickel-iron battery. the following reaction takes place in the battery. fe(s) + 2 nio(oh)(s) + 2 h2o(l) fe(oh)2(s) + 2 ni(oh)2(aq) how many mole of fe(oh)2, is produced when 4.20 mol fe and 6.70 mol nio(oh) react?
Answers: 3

Chemistry, 23.06.2019 07:00
An unknown substance is a white solid at room temperature and has a melting point of 78 °c. which of the following substances is most likely to be the identity of the unknown sample?
Answers: 1
You know the right answer?
If the half-life of 37rb is 4.7x101 years, how long would it take for 0.5 grams of a 2 gram sample t...
Questions

Biology, 13.01.2021 21:20

Mathematics, 13.01.2021 21:20



Physics, 13.01.2021 21:20


English, 13.01.2021 21:20

English, 13.01.2021 21:20



Mathematics, 13.01.2021 21:20




Biology, 13.01.2021 21:20

Mathematics, 13.01.2021 21:20

History, 13.01.2021 21:20



Mathematics, 13.01.2021 21:20

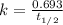
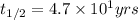
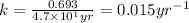
![k=\frac{2.303}{t}\log\frac{[A_o]}{[A]}](/tpl/images/0258/8865/f1041.png)
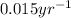
![[A_o]](/tpl/images/0258/8865/dc622.png) = initial amount of the reactant = 2 g
= initial amount of the reactant = 2 g