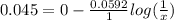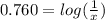Chemistry, 26.11.2019 04:31 samsmith666
Asilver nitrate, agno3, solution of unknown concentration was discovered in the lab. to determine the concentration of the solution, a concentration cell was set up with the unknown solution in the anode compartment and a 1.0 m agno3 solution in the cathode compartment. the cell had a potential (e) of +0.045 v at 25°c. what is the concentration of silver in the unknown solution?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 05:00
You mix the pks of succinic acid are 4.21 and 5.64. how many gramsa graduate student at sdsu wants to measure the activity of a particular enzyme at ph 4.0. to buffer her reaction, she will use a buffer system based on one of the acids listed below, which acid is most appropriate for the experiment? of monosodium succinate (fw = 140 g/mol) and disodium succinate (fw = 162 g/mol) must be added to 1 l of water to produce a solution with a ph 5.28 and a total solute concentration of 100 mm? (assume the total volume remains 1 liter, answer in grams monosodium succinate, grams disodium succinate, respectively.) volumes of 0.05 m nah2po4 and 0.05 m na2hpo4 (pk's for phosphoric acid are 2.15, 6.82 and 12.38). which of the following best describes the resulting solution?
Answers: 2

Chemistry, 22.06.2019 09:40
How many grams of aluminum will there be in 98g of al2o3?
Answers: 1

Chemistry, 22.06.2019 10:00
Miner's coal distributors does not mine coal itself, nor does it even store or handle the coal. instead, miner's solicits orders for low sulfur coal from other firms, then purchases the required amount from suppliers and directs them to ship the coal to its customers. what is miner's
Answers: 1

You know the right answer?
Asilver nitrate, agno3, solution of unknown concentration was discovered in the lab. to determine th...
Questions

Mathematics, 30.03.2020 23:53








Mathematics, 30.03.2020 23:53

Mathematics, 30.03.2020 23:53



Computers and Technology, 30.03.2020 23:53







![Ecell=E^{0}_{cell}-\frac{0.0592}{n}log\frac{[anodic]}{[cathodic]}](/tpl/images/0391/0151/f32d1.png)