
Chemistry, 30.11.2019 07:31 sophiateaches053
When a 3.23 g sample of solid sodium hydroxide was dissolved in a calorimeter in 100.0 g of water, the temperature rose from 23.9 °c to 32.0 °c. calculate δh (in kj/mol naoh) for the following solution process: naoh(s) →na+(aq)+ oh−(aq)

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 19:30
Motivation cannot be developed with practice; a person either possesses it or they do not.
Answers: 1

Chemistry, 22.06.2019 00:30
Maria wants to determine which type of disinfectant kills the most bacteria. which of the following is the best way for maria to determine this? a. ask ten different companies that make disinfectants which type is best. b. put the same amount and species of bacteria on ten identical plates, and add ten different kinds of disinfectant to each plate. c. interview ten different people to determine which type of disinfectant they prefer. d. put the same amount and species of bacteria on ten identical plates, and add a different disinfectant to each plate.
Answers: 1

Chemistry, 22.06.2019 10:00
Suppose the universe were completely empty except for one object-a solid sphere moving through space of 100 km/s. what sort of path would the object be moving in? explain your answer
Answers: 1

Chemistry, 22.06.2019 12:30
According to the valence shell electron pair repulsion (vsepr) theory, a molecule that has four electron groups around the central atom will exhibit what electron geometry? view available hint(s) according to the valence shell electron pair repulsion (vsepr) theory, a molecule that has four electron groups around the central atom will exhibit what electron geometry? trigonal bipyramidal tetrahedral square planar determination of electron geometry requires information on whether the electron groups are lone pairs or bonding groups.
Answers: 2
You know the right answer?
When a 3.23 g sample of solid sodium hydroxide was dissolved in a calorimeter in 100.0 g of water, t...
Questions


Mathematics, 08.11.2020 14:00

Health, 08.11.2020 14:00


Mathematics, 08.11.2020 14:00

Physics, 08.11.2020 14:00

Mathematics, 08.11.2020 14:00


English, 08.11.2020 14:00

Chemistry, 08.11.2020 14:00

English, 08.11.2020 14:00

Mathematics, 08.11.2020 14:00

Business, 08.11.2020 14:00


English, 08.11.2020 14:00

English, 08.11.2020 14:00


History, 08.11.2020 14:00


Mathematics, 08.11.2020 14:00

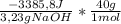 = -41929J/molNaOH ≡
= -41929J/molNaOH ≡ 

