Chemistry, 19.12.2019 01:31 asiababbie33
The al2o3 crystal structure (corundum) consists of an hcp arrangement of o2- ions; the al3+ ions occupy octahedral positions. what fraction of the available octahedral positions are filled with al3+ ions? in corundum, the al3+ ions do not fill the tetrahedral positions.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 12:10
Consider the reaction: n2(g) + o2(g) ⇄ 2no(g) kc = 0.10 at 2000oc starting with initial concentrations of 0.040 mol/l of n2 and 0.040 mol/l of o2, calculate the equilibrium concentration of no in mol/l how would this be done?
Answers: 3

Chemistry, 22.06.2019 16:10
Predict the reactants of this chemical reaction. that is, fill in the left side of the chemical equation. be sure the equation you submit is balanced. (you can edit both sides of the equation to balance it, if you need to.) note: you are writing the molecular, and not the net ionic equation. > cacl2(aq) + h20(l)
Answers: 2

Chemistry, 22.06.2019 19:00
What information does a complete ionic equation give that the balanced equation doesn’t show?
Answers: 1

Chemistry, 23.06.2019 01:00
Which of the following is in the lanthanide family? a) uranium b) promethium c) silver d) gold
Answers: 2
You know the right answer?
The al2o3 crystal structure (corundum) consists of an hcp arrangement of o2- ions; the al3+ ions oc...
Questions




Mathematics, 08.10.2019 03:00

Mathematics, 08.10.2019 03:00


Mathematics, 08.10.2019 03:00

Mathematics, 08.10.2019 03:00



Mathematics, 08.10.2019 03:00

Mathematics, 08.10.2019 03:00

Chemistry, 08.10.2019 03:00

Mathematics, 08.10.2019 03:00




Mathematics, 08.10.2019 03:00

Mathematics, 08.10.2019 03:00

Mathematics, 08.10.2019 03:00

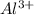 ions is
ions is 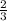
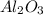 crystal structure consists of an HCP arrangement of
crystal structure consists of an HCP arrangement of 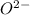 ions.
ions.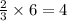
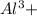 ions are present = 4
ions are present = 4