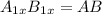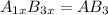Chemistry, 19.12.2019 20:31 clmorcutt420
In a hexagonal-close-pack (hcp) unit cell, the ratio of lattice points to octahedral holes to tetrahedral holes is 1: 1: 2.
what is the general formula for a compound if anions occupy the hcp lattice points and cations occupy all the octahedral holes? what is the general formula for a compound if anions occupy the hcp lattice points and cations occupy all the octahedral and tetrahedral holes?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 04:40
Silver tarnishes as silver metal reacts with hydrogen sulfide, h2s, in the air. in this reaction, dark silver sulfide, au2s, covers the surface of silver. when silver is polished, this coating of silver sulfide can be removed from the surface. this makes the silver shiny again. enter the coefficients that balance the tarnishing reaction equation. (type 1 for no coefficient.)
Answers: 2

Chemistry, 22.06.2019 10:30
Which of these is not an example of chemical weathering? a. iron-rich mineral rusting b. feldspar turning into clay c. limestone reacting with acid d. granite breaking up into sand
Answers: 1

Chemistry, 22.06.2019 20:00
Suppose that some of the compound spilled out of the crucible after it was heated. would that cause the percent by mass of water in the compound determined by the experiment to be too low, too high, or unchanged? briefly explain your answer.
Answers: 1

Chemistry, 22.06.2019 22:30
Vi limitens. vastery test select the correct answer. which statement explains why large atoms are more reactive than small atoms? a. large atoms have valence electrons farther from the nucleus and lose them more readily. b. large atoms have greater ionization energy, which they can utilize during a reaction. c. large atoms have a greater number of electrons that they can lose during a reaction. d. large atoms have more energy levels, so they have more energy to pass on in a reaction. reset next
Answers: 3
You know the right answer?
In a hexagonal-close-pack (hcp) unit cell, the ratio of lattice points to octahedral holes to tetrah...
Questions

Mathematics, 22.07.2019 20:00

Physics, 22.07.2019 20:00

Biology, 22.07.2019 20:00

Social Studies, 22.07.2019 20:00

Mathematics, 22.07.2019 20:00

Biology, 22.07.2019 20:00


History, 22.07.2019 20:00

Mathematics, 22.07.2019 20:00

Social Studies, 22.07.2019 20:00



History, 22.07.2019 20:00

Social Studies, 22.07.2019 20:00

Social Studies, 22.07.2019 20:00