Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 00:00
Select the correct answer. which statement is true about a polarized object? o a. it gains electrons and becomes negatively charged. ob. it gains protons and becomes positively charged. oc. the number of positive and negative charges can be the same. od. it has to be a metal. o e. there is no change in the distribution of the charge in the object. reset next what
Answers: 3

Chemistry, 22.06.2019 09:00
Identify the electromagnets with poles that are reversed from the electromagnet shown above
Answers: 3

Chemistry, 22.06.2019 16:00
Sulfuric acid is a polyprotic acid. write balanced chemical equations for the sequence of reactions that sulfuric acid can undergo when it's dissolved in water.
Answers: 2

Chemistry, 22.06.2019 16:30
How many grams of mgbr2 are needed to produce 75g or metal?
Answers: 1
You know the right answer?
In a reaction, gaseous reactants form a liquid product. The heat absorbed by the surroundings is 1.1...
Questions

Mathematics, 13.12.2019 05:31


Mathematics, 13.12.2019 05:31



Mathematics, 13.12.2019 05:31


Mathematics, 13.12.2019 05:31

Mathematics, 13.12.2019 05:31


Mathematics, 13.12.2019 05:31

Mathematics, 13.12.2019 05:31


Computers and Technology, 13.12.2019 05:31

Mathematics, 13.12.2019 05:31

Mathematics, 13.12.2019 05:31

Advanced Placement (AP), 13.12.2019 05:31

Mathematics, 13.12.2019 05:31


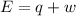
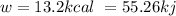
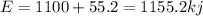
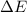 is 1155 kJ
is 1155 kJ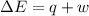
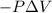 {Work done on the system is positive as the final volume is lesser than initial volume}
{Work done on the system is positive as the final volume is lesser than initial volume}
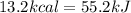 (1kcal=4.184 kJ)
(1kcal=4.184 kJ)