

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 12:30
Activity two: just lemons, inc. production here's a one-batch sample of just lemons lemonade production. determine the percent yield and amount of leftover ingredients for lemonade production and place your answers in the data chart. hint: complete stoichiometry calculations for each ingredient to determine the theoretical yield. complete a limiting reactant-to-excess reactant calculation for both excess ingredients. water sugar lemon juice lemonade percent yield leftover ingredients 946.36 g 196.86 g 193.37 g 2050.25 g just lemons lemonade recipe equation: 2 water + sugar + lemon juice = 4 lemonade mole conversion factors: 1 mole of water = 1 cup = 236.59 g 1 mole of sugar = 1 cup = 225 g 1 mole of lemon juice = 1 cup = 257.83 g 1 mole of lemonade = 1 cup = 719.42 g
Answers: 2

Chemistry, 21.06.2019 18:00
Acylinder is filled with 2.00 moles of nitrogen, 3.00 moles of argon and 5.00 moles of helium. if the gas mixture is at stp, what is the partial pressure of the argon
Answers: 1


Chemistry, 21.06.2019 22:30
Ibeg i need 20. a reaction produces 4.93 l of oxygen, but was supposed to produce 1 mol of oxygen. what is the percent yield?
Answers: 1
You know the right answer?
Use values of Ksp for AgI and Kf for Ag(CN)2− to calculate the molar solubility of AgI in pure water...
Questions

Mathematics, 05.11.2020 17:20







Mathematics, 05.11.2020 17:20

Arts, 05.11.2020 17:20







Physics, 05.11.2020 17:20

English, 05.11.2020 17:20

Arts, 05.11.2020 17:20


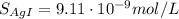
![S_{[ AgI]} = 0.05 M](/tpl/images/0550/2696/fce57.png)
![K_{sp} = [Ag^{+}][I^{-}] = 8.3\cdot 10^{-17}](/tpl/images/0550/2696/5785c.png)
![K_{sp} = [Ag^{+}]^{2} \rightarrow S = \sqrt{K_{sp}} = \sqrt{8.3\cdot 10^{-17}} = 9.11 \cdot 10^{-9} mol/L](/tpl/images/0550/2696/2ba74.png)
![K_{sp} = [Ag^{+}][I^{-}]](/tpl/images/0550/2696/d6ef6.png) (2)
(2)![K_{f} = \frac{[Ag(CN)_{2}^{-}]}{[Ag^{+}][CN^{-}]^{2}}](/tpl/images/0550/2696/5bfe2.png) (4)
(4)![K_{eq} = \frac{[Ag(CN)_{2}^{-}][I^{-}]}{[CN^{-}]^{2}}](/tpl/images/0550/2696/ca1d7.png) (6)
(6)![[I^{-}] = \frac{K_{sp}}{[Ag^{+}]}](/tpl/images/0550/2696/b9a73.png) (7)
(7)![K_{eq} = \frac{[Ag(CN)_{2}^{-}]}{[CN^{-}]^{2}}*\frac{K_{sp}}{[Ag^{+}]}](/tpl/images/0550/2696/45445.png)
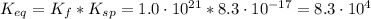
![K_{eq} = \frac{[Ag(CN)_{2}^{-}][I^{-}]}{[CN^{-}]^{2}} = \frac{x*x}{(0.1 - 2x)^{2}}](/tpl/images/0550/2696/83c45.png)
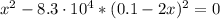
![x = 0.05 mol/L = S_{[ AgI]}](/tpl/images/0550/2696/daf71.png)


