
Chemistry, 23.03.2020 21:11 copelandgarret9972
What concentration of Cl2O remains after a mixture that initially contains [H2O] = 1.00 M and [Cl2O] = 1.00 M comes to equilibrium at 25 °C ? Kc for the reaction is 0.0900.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 17:50
An aqueous solution of potassium hydroxide is standardized by titration with a 0.194 m solution of hydrobromic acid. if 25.2 ml of base are required to neutralize 24.2 ml of the acid, what is the molarity of the potassium hydroxide solution? m potassium hydroxide
Answers: 2


Chemistry, 23.06.2019 03:30
Ineed pls urgent 1-20 in order and fully detail step my step.
Answers: 1

Chemistry, 23.06.2019 04:40
6) (a) calculate the absorbance of the solution if its concentration is 0.0278 m and its molar extinction coefficient is 35.9 l/(mol cm). the depth of the cell is 5 mm. (b) what is the %t? (7) calculate the absorbance of the solution if the transmitted light intensity is 70% of the initial light beam intensity
Answers: 1
You know the right answer?
What concentration of Cl2O remains after a mixture that initially contains [H2O] = 1.00 M and [Cl2O]...
Questions


English, 22.09.2019 09:00

Mathematics, 22.09.2019 09:00



Mathematics, 22.09.2019 09:00





Mathematics, 22.09.2019 09:00



Geography, 22.09.2019 09:00

Biology, 22.09.2019 09:00

Computers and Technology, 22.09.2019 09:00

Social Studies, 22.09.2019 09:00


Mathematics, 22.09.2019 09:00

Mathematics, 22.09.2019 09:00

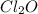 is 0.870 M.
is 0.870 M.![[H_2O]=1.00 M](/tpl/images/0559/4667/b0041.png)
![Cl_2O=[Cl_2O]=1.00 M](/tpl/images/0559/4667/4c14a.png)
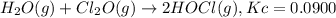
![K_c=\frac{[HOCl]^2}{[H_2O][Cl_2O]}](/tpl/images/0559/4667/da783.png)
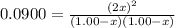
![[Cl_2O]=(1,00-x) M=1.00 M-0.130 M=0.870 M](/tpl/images/0559/4667/c9bd4.png)



