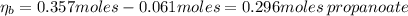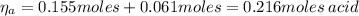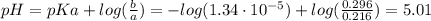A 1.41 L buffer solution consists of 0.253 M propanoic acid and 0.110 M sodium propanoate. Calculate the pH of the solution following the addition of 0.061 mol HCl . Assume that any contribution of the HCl to the volume of the solution is negligible. The K a of propanoic acid is 1.34 × 10 − 5 .

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 20:40
You may expect bonds between two atoms which each have n covalent lonic metallic hydrogen
Answers: 2

Chemistry, 22.06.2019 11:50
If oil spills continue, all of the following should be expected except (2 points) death of aquatic life. polluted groundwater. decreased soil productivity. increased global temperatures.
Answers: 3

Chemistry, 22.06.2019 19:00
What is the compound name for the formula [ru(en)2cl2]2+ and [co(en)cl2br]-
Answers: 1

Chemistry, 22.06.2019 23:00
In the reaction h2co3 (aq) + 3nh3 (aq) = 2 nh4+ (aq) + co3 2-, how many electrons are transferred?
Answers: 3
You know the right answer?
A 1.41 L buffer solution consists of 0.253 M propanoic acid and 0.110 M sodium propanoate. Calculate...
Questions

English, 23.10.2019 22:10



Mathematics, 23.10.2019 22:10


Mathematics, 23.10.2019 22:10

Biology, 23.10.2019 22:10


Mathematics, 23.10.2019 22:10

History, 23.10.2019 22:10





Social Studies, 23.10.2019 22:10


Biology, 23.10.2019 22:10

English, 23.10.2019 22:10

Business, 23.10.2019 22:10

Chemistry, 23.10.2019 22:10

![\eta_{a} = [a]*Va = 0.110 M * 1.41 L = 0.155 moles \thinspace acid](/tpl/images/0566/2281/d601e.png)
![\eta_{b} = [b]*Vb = 0.253 M * 1.41 L = 0.357 moles\thinspace propanoate](/tpl/images/0566/2281/b75d2.png)