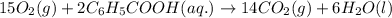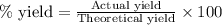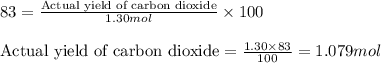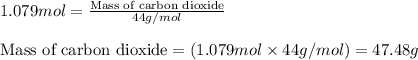Chemistry, 30.03.2020 20:24 gibbss80stu
Consider the following balanced equation for the following reaction:
15O2(g) + 2C6H5COOH(aq) → 14CO2(g) + 6H2O(l)
Determine the amount of CO2(g) formed in the reaction if the percent yield of CO2(g) is 83.0% and the theoretical yield of CO2(g) is 1.30 moles.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 16:40
Let the ed50 of a recreational drug be defined as the amount required for 50% of a test group to feel high or get a buzz. if the ed50 value of ethanol is 470 mg/kg body mass, what dose would a 70 kg party goer need to quickly consume in order to have a 50% chance of getting a buzz? 235 mg 470 mg 32,900 mg 35,000,000 mg
Answers: 3

Chemistry, 22.06.2019 20:50
What is the vapor pressure of a solution with a benzene to octane?
Answers: 2

Chemistry, 23.06.2019 11:30
A) equal lines b) parallel lines c) perpendicular lines d) none of the above
Answers: 1

Chemistry, 23.06.2019 18:10
Which is an aspect of the kinetic-molecular theory and can be used to explain the behavior of plasmas? particle spacing can allow a very high density. particle kinetic energy is independent of temperature. particles vibrate quickly in stationary positions. particles exchange energy through elastic collisions.
Answers: 2
You know the right answer?
Consider the following balanced equation for the following reaction:
15O2(g) + 2C6H5COOH(aq) →...
15O2(g) + 2C6H5COOH(aq) →...
Questions


Mathematics, 20.02.2021 15:10

Chemistry, 20.02.2021 15:10

Mathematics, 20.02.2021 15:10

Computers and Technology, 20.02.2021 15:10

Mathematics, 20.02.2021 15:10

Mathematics, 20.02.2021 15:10

English, 20.02.2021 15:10


Physics, 20.02.2021 15:10



Mathematics, 20.02.2021 15:10

Mathematics, 20.02.2021 15:10


Arts, 20.02.2021 15:10

Arts, 20.02.2021 15:10

English, 20.02.2021 15:10

History, 20.02.2021 15:20

Mathematics, 20.02.2021 15:20