
For the reaction H2(g) + I2(g) 2HI(g), Kc = 50.2 at 445ºC. If [H2] = [I2] = [HI] = 1.75 10-3 M at 445ºC, which one of these statements is true? A) The system is at equilibrium, thus no concentration changes will occur. B) The concentrations of HI and I2 will increase as the system approaches equilibrium. C) The concentration of HI will increase as the system approaches equilibrium. D) The concentrations of H2 and HI will fall as the system moves toward equilibrium. E) The concentrations of H2 and I2 will increase as the system approaches equilibrium

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 03:30
In saturated organic compounds, all the bonds between carbon atoms are called?
Answers: 1

Chemistry, 22.06.2019 22:30
Amedication is given at a dosage of 3.000 mg of medication per kg of body weight. if 0.1500 g of medication is given, then what was the patient's weight in pounds (lbs)? there are 453.59g in 1 lb.
Answers: 2

Chemistry, 23.06.2019 02:00
The point along a planet's orbit where it is closest to the sun is called the
Answers: 1

Chemistry, 23.06.2019 06:30
Which of the following steps is not likely to take place during cellular respiration? (5 points) select one: a. oxygen combines with carbon of simple sugar. b. energy molecule transfers energy to cells. c. simple sugar breaks down. d. energy is used up.
Answers: 1
You know the right answer?
For the reaction H2(g) + I2(g) 2HI(g), Kc = 50.2 at 445ºC. If [H2] = [I2] = [HI] = 1.75 10-3 M at...
Questions



Mathematics, 23.07.2021 14:00

Social Studies, 23.07.2021 14:00

Mathematics, 23.07.2021 14:00



History, 23.07.2021 14:00

Mathematics, 23.07.2021 14:00

Mathematics, 23.07.2021 14:00

Computers and Technology, 23.07.2021 14:00

History, 23.07.2021 14:00

Mathematics, 23.07.2021 14:00


Chemistry, 23.07.2021 14:00


English, 23.07.2021 14:00

Law, 23.07.2021 14:00

Mathematics, 23.07.2021 14:00

Mathematics, 23.07.2021 14:00

 is written as:
is written as:![Q_c=\frac{[HI]^2}{[H_2]^1[I_2]^1}](/tpl/images/0575/8711/8578e.png)
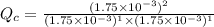
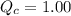
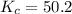
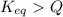 , the reaction will shift towards the right i.e. towards the product side.
, the reaction will shift towards the right i.e. towards the product side.

