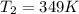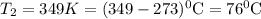Mount Everest rises to a height of 8,850 m above sea level. At a base camp on the mountain the atmospheric pressure is measured to be 314.0 mm Hg. At what temperature (in °C) will water boil at base camp ? The vapor pressure of water at 373 K is 760.0 mm Hg. (ΔH°vap for H2O = 40.7 kJ/mol and R = 8.314 J/mol K).
a. 344°C.
b. 70.8°C .
c. 2.91E-3°C .
d. 79.8°C.
e. 57.8°C.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 17:30
What is the percentage by mass of silicon (si) in iron aluminum silicate (fe3al2(sio4)3)?
Answers: 2

Chemistry, 22.06.2019 11:00
Iron (3) oxide will decompose in the presence of hydrogen gas and heater to produced iron and digydrogen monoxide white a balanced chemical equation
Answers: 1


Chemistry, 23.06.2019 02:00
Which would freeze at a higher temperature: the great salt lake or lake tahoe? a. lake tahoe would freeze at a higher temperature. b. the great salt lake would freeze at a higher temperature. c. both lakes would freeze at the same temperature.
Answers: 2
You know the right answer?
Mount Everest rises to a height of 8,850 m above sea level. At a base camp on the mountain the atmos...
Questions


Computers and Technology, 09.10.2019 16:20


Health, 09.10.2019 16:20

Mathematics, 09.10.2019 16:20

Mathematics, 09.10.2019 16:20

History, 09.10.2019 16:20


Mathematics, 09.10.2019 16:20




Mathematics, 09.10.2019 16:20


Mathematics, 09.10.2019 16:20

History, 09.10.2019 16:20

World Languages, 09.10.2019 16:20


Mathematics, 09.10.2019 16:20

SAT, 09.10.2019 16:20

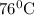 .
.![ln(\frac{P_{2}}{P_{1}})=\frac{-\Delta H_{vap}^{0}}{R}[\frac{1}{T_{2}}-\frac{1}{T_{1}}]](/tpl/images/0599/0769/06906.png)
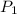 and
and 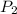 are vapor pressures of liquid at
are vapor pressures of liquid at 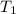 (in kelvin) and
(in kelvin) and 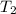 (in kelvin) temperatures respectively.
(in kelvin) temperatures respectively.![ln(\frac{314.0}{760.0})=\frac{-40.7\times 10^{3}\frac{J}{mol}}{8.314\frac{J}{mol.K}}\times [\frac{1}{T_{2}}-\frac{1}{373K}]](/tpl/images/0599/0769/811c1.png)