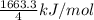Chemistry, 15.04.2020 02:05 melissakm77
Given that Δ H ∘ f [ H ( g ) ] = 218.0 kJ ⋅ mol − 1 Δ H ∘ f [ C ( g ) ] = 716.7 kJ ⋅ mol − 1 Δ H ∘ f [ CH 4 ( g ) ] = − 74.6 kJ ⋅ mol − 1 calculate the average molar bond enthalpy of the carbon‑hydrogen bond in a CH 4 molecule

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 00:00
How many liters of water vapor can be produced if 108 grams of methane gas (ch4) are combusted at 312 k and 0.98 atm? show all work. pls ! will mark as brainliest
Answers: 1

Chemistry, 22.06.2019 14:30
The three types is stress that act on earths rocks are compression, tension, and
Answers: 1

Chemistry, 22.06.2019 16:00
He table below gives the atomic mass and relative abundance values for the three isotopes of element m. relative abundance (%) atomic mass (amu) 78.99 23.9850 10.00 24.9858 11.01 25.9826 what is the average atomic mass (in amu) of element m? 2.86 5.36 24.30 24.98
Answers: 2

Chemistry, 22.06.2019 20:30
Some familiar products contain some of the same types of atoms. for instance, the chemical formula for baking soda is nahco 3. the chemical formula for liquid bleach is naclo, and the chemical formula for table salt is nacl. which choice best describes why these three products have some of the same types of atoms in common?
Answers: 1
You know the right answer?
Given that Δ H ∘ f [ H ( g ) ] = 218.0 kJ ⋅ mol − 1 Δ H ∘ f [ C ( g ) ] = 716.7 kJ ⋅ mol − 1 Δ H ∘ f...
Questions

Biology, 13.10.2020 18:01



Mathematics, 13.10.2020 18:01


Mathematics, 13.10.2020 18:01

Mathematics, 13.10.2020 18:01


Social Studies, 13.10.2020 18:01


English, 13.10.2020 18:01



Computers and Technology, 13.10.2020 18:01

Biology, 13.10.2020 18:01


Mathematics, 13.10.2020 18:01


Geography, 13.10.2020 18:01

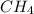 is 415.825 kJ/mol.
is 415.825 kJ/mol.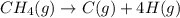
![\Delta H^{0}=[1mol\times \Delta H_{f}^{0}(C)_{g}]+[4mol\times \Delta H_{f}^{0}(H)_{g}]-[1mol\times \Delta H_{f}^{0}(CH_{4})_{g}]](/tpl/images/0600/7413/0c0db.png)
![[1mol\times 716.7\frac{kJ}{mol}]+[4mol\times 218.0\frac{kJ}{mol}]-[1mol\times -74.6\frac{kJ}{mol}]=1663.3kJ](/tpl/images/0600/7413/961ee.png)