Chemistry, 22.04.2020 20:04 SMURFETTE86
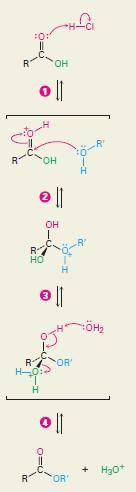
Esters can be synthesized by an acid-catalyzed nucleophilic acyl substitution between an alcohol and a carboxylic acid; this process is called the Fischer esterification reaction. Because the alcohol oxygen is a poor nucleophile, the carbonyl carbon is made a better electrophile by protonation of the carbonyl oxygen. The steps of the synthesis are all reversible. The reaction is generally driven to completion by using an excess of the liquid alcohol as a solvent, or by distilling off the product as it forms. Draw curved arrows to show the movement of electrons in this step of the mechanism.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 06:10
How many moles of gas are present if p=11 atm, v=12l, t=185k?
Answers: 1

Chemistry, 22.06.2019 10:00
Drug abuse will not lead to physical and psychological dependence. true or false ?
Answers: 2

Chemistry, 22.06.2019 14:30
For the reaction shown, find the limiting reactant for each of the following initial amounts of reactants. 4al(s)+3o2(g)→2al2o3(s) a) 1 molal, 1 mol o2 b) 4 molal, 2.6 mol o2 c) 16 molal, 13 mol o2 d) 7.4 molal, 6.5 mol o2
Answers: 3

You know the right answer?
Esters can be synthesized by an acid-catalyzed nucleophilic acyl substitution between an alcohol and...
Questions

History, 17.03.2020 01:08


Health, 17.03.2020 01:08



Mathematics, 17.03.2020 01:09

History, 17.03.2020 01:09

Mathematics, 17.03.2020 01:09




Mathematics, 17.03.2020 01:09

Mathematics, 17.03.2020 01:09




Mathematics, 17.03.2020 01:09