
Chemistry, 29.05.2020 06:02 angelZ3947
The first two steps in the industrial synthesis of nitric acid produce nitrogen dioxide from ammonia: The net reaction is: Write an equation that gives the overall equilibrium constant in terms of the equilibrium constants and . If you need to include any physical constants, be sure you use their standard symbols, which you'll find in the ALEKS Calculator.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 22:30
For the following, determine the type of reaction and then give products.
Answers: 2

Chemistry, 22.06.2019 06:00
An atom of sodium-23 (atomic number = 11) has a positive charge of +1. give this information, how many electrons does it have? how many proteins and neutrons does this atom have
Answers: 2

Chemistry, 22.06.2019 16:40
The diagram below shows the movement of particles. what does this piece of evidence best support? the collision theory the maxwell-boltzmann distribution the effect of pressure on reaction rates the effect of temperature on reaction rates
Answers: 3

Chemistry, 22.06.2019 20:00
Carbon-14 undergoes radioactive decay in the reaction above. determine the type of radiation emitted in this reaction and describe what is happening to the nucleus during this reaction.
Answers: 2
You know the right answer?
The first two steps in the industrial synthesis of nitric acid produce nitrogen dioxide from ammonia...
Questions


Mathematics, 16.11.2019 09:31








Social Studies, 16.11.2019 09:31


Physics, 16.11.2019 09:31

History, 16.11.2019 09:31



Biology, 16.11.2019 09:31

Mathematics, 16.11.2019 09:31

History, 16.11.2019 09:31


Social Studies, 16.11.2019 09:31

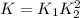
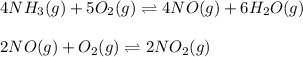
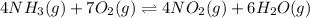
![K_1=\frac{[NO]^4[H_2O]^6}{[NH_3]^4[O_2]^5}\\ \\K_2=\frac{[NO_2]^2}{[NO]^2[O_2]}](/tpl/images/0669/9771/004ed.png)
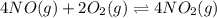
![K_2^{new}=\frac{[NO_2]^4}{[NO]^4[O_2]^2}](/tpl/images/0669/9771/b0ba1.png)
![K_1*K_2^2=\frac{[NO]^4[H_2O]^6}{[NH_3]^4[O_2]^5}*\frac{[NO_2]^4}{[NO]^4[O_2]^2}=\frac{[H_2O]^6[NO_2]^4}{[NH_3]^4[O_2]^7}](/tpl/images/0669/9771/5162f.png)
![K=\frac{[H_2O]^6[NO_2]^4}{[NH_3]^4[O_2]^7}](/tpl/images/0669/9771/dc773.png)


