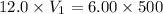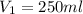Chemistry, 05.06.2020 08:59 rmcarde3453
You are required to prepare 500 ml of a 6.00 M solution of HNO3 from a stock solution of 12.0 M. Describe in detail how you would go about preparing this solution. Clearly state the volume of stock solution used, the glassware's used and the procedure.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 19:00
Agood hypothesis includes which of the following? a: prediction b: data c: uncertainty d: conclusion
Answers: 1

Chemistry, 21.06.2019 22:30
Will mark brainliest 26. which of these statements are true? (3 points) a. gases are compressible b. gases fill their containers completely c. the pressure of a gas is independent of the temperature d. gases have mass e. gases exert pressure f. the pressure of a gas is dependent on the volume g. gas pressure results from the collisions between gas particles h. gases have a definite volume and shape
Answers: 1

Chemistry, 22.06.2019 00:30
Elements that do not have full outer electron shells will donate, share, or take electrons from other atoms. choose the items that have the correct binary ionic formula.
Answers: 2

Chemistry, 23.06.2019 08:00
What is the temperature in kelvin of a gas if it is allowed to expand from 1.50 l to 4.50 l? the initial temperature is 10.0°c and pressure is constant throughout the change. which equation should you use? t2= v2/v1 t1 what is the final temperature? ⇒ 849 k these are the answers.
Answers: 1
You know the right answer?
You are required to prepare 500 ml of a 6.00 M solution of HNO3 from a stock solution of 12.0 M. Des...
Questions

History, 23.05.2020 17:00







Mathematics, 23.05.2020 17:00





Computers and Technology, 23.05.2020 17:00







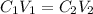
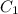 = concentration of stock solution = 12.0 M
= concentration of stock solution = 12.0 M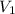 = volume of stock solution = ?
= volume of stock solution = ?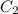 = concentration of diluted solution= 6.00 M
= concentration of diluted solution= 6.00 M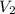 = volume of diluted acid solution = 500 ml
= volume of diluted acid solution = 500 ml