
The equilibrium constant for the reaction NO2(g)+NO3(g)→N2O5(g) is 2.1x10-20 , therefore: a. At equilibrium, the concentration of products and reactants is about the same. b. At equilibrium, the concentration of products is greater than the reactants. c. At equilibrium, the concentration of reactants is greater than the products

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 15:00
Which of the following elements is a representative element? a. chromium (cr) b. aluminum (al) c. mercury (hg) d. silver (ag)
Answers: 3

Chemistry, 21.06.2019 20:20
Calculate the molarity of the solution. 6.02 x 1022 molecules of hci (molecular weight = 36.5 g/mole) in 2.0 liters of water m
Answers: 1

Chemistry, 22.06.2019 03:40
Chemical kinetics what was the rate of reaction in trial 3? choose the closest answer.
Answers: 3

Chemistry, 22.06.2019 06:00
Compare and contrast physical changes with chemical changes.
Answers: 1
You know the right answer?
The equilibrium constant for the reaction NO2(g)+NO3(g)→N2O5(g) is 2.1x10-20 , therefore: a. At equ...
Questions



Mathematics, 12.07.2019 20:20



Mathematics, 12.07.2019 20:20





Mathematics, 12.07.2019 20:20




Computers and Technology, 12.07.2019 20:20





Mathematics, 12.07.2019 20:20

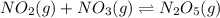
![K_{eq}=\frac{[N_2O_5]}{[NO_2]\times [NO_3]}](/tpl/images/0694/4070/44e17.png)
![2.1\times 10^{-20}=\frac{[N_2O_5]}{[NO_2]\times [NO_3]}](/tpl/images/0694/4070/ede48.png)
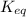 is
is 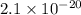 which is less than 1,
which is less than 1, 

