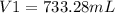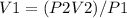Chemistry, 27.06.2020 17:01 wolfking800
A sample of chlorine gas starting at 686 mm Hg is placed under a pressure of 991 mm Hg and reduced to a volume of 507.6 mL. What was the initial volume of the chlorine gas container if the process was performed at constant temperature

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 14:00
Calculate the energy required to ionize a hydrogen atom to an excited state where the electron is initially in the n = 5 energy level. report your answer in kilojoules
Answers: 1

Chemistry, 23.06.2019 07:00
An unknown substance is a white solid at room temperature and has a melting point of 78 °c. which of the following substances is most likely to be the identity of the unknown sample? a. naphthalene, a molecular solid with the formula c10h8 b. silica, a network solid held together by covalent bonds with the formula sio2 c. calcium chloride, an ionic compound with the formula cacl2 d. water, an molecular compound with the formula h2o
Answers: 2

Chemistry, 23.06.2019 11:50
Charles's law describes the relationship of the volume and temperature of gas at a constant mass and pressure. according to this law, what would happen to the temperature of the gas if its volume decreased from 1.0 l to 0.50 l?
Answers: 3

Chemistry, 23.06.2019 16:30
Ashell can hold a maximum of 32 electrons, what is the value of n?
Answers: 3
You know the right answer?
A sample of chlorine gas starting at 686 mm Hg is placed under a pressure of 991 mm Hg and reduced t...
Questions

Business, 12.01.2021 01:00


English, 12.01.2021 01:00

Social Studies, 12.01.2021 01:00

Spanish, 12.01.2021 01:00

Computers and Technology, 12.01.2021 01:00




Mathematics, 12.01.2021 01:00

Chemistry, 12.01.2021 01:00


Geography, 12.01.2021 01:00


Mathematics, 12.01.2021 01:00

Computers and Technology, 12.01.2021 01:00

Mathematics, 12.01.2021 01:00


Chemistry, 12.01.2021 01:00

Chemistry, 12.01.2021 01:00