
Chemistry, 14.07.2020 01:01 eggemotions
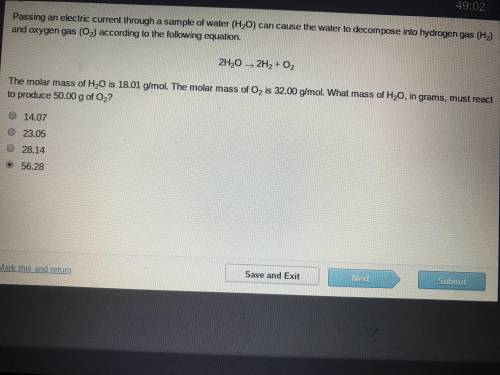
The molar mass of H2O is 18.01 g/mol. The molar mass of O2 is 32.00 g/mol. What mass of H2O, ins grams, must react to produce 50.00 g of O2

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 01:30
Phosphorous acid, h3po3(aq) , is a diprotic oxyacid that is an important compound in industry and agriculture. the values of phosphorous acid are 1.30 6.70 calculate the ph for each of the given points in the titration of 50.0 ml of 1.5 m h3po3(aq) with 1.5 m koh(aq) .
Answers: 3

Chemistry, 22.06.2019 03:10
The covalent compound acetylene, which is the fuel of the oxyacetylene torch used by welders, has the molecular formula c2h2. the covalent compound benzene, a commercial solvent, has the molecular formula c6h6 each of these covalent compounds contains carbon and hydrogen atoms in a one-to-one ratio. would it be correct to write the chemical formulas of each as ch? explain.
Answers: 1


Chemistry, 23.06.2019 11:30
How do you calculate the mass of a product when the amounts of more than one reactant are given?
Answers: 3
You know the right answer?
The molar mass of H2O is 18.01 g/mol. The molar mass of O2 is 32.00 g/mol. What mass of H2O, ins gra...
Questions

History, 06.12.2019 13:31

Mathematics, 06.12.2019 13:31

Health, 06.12.2019 13:31

Mathematics, 06.12.2019 13:31



English, 06.12.2019 14:31

Biology, 06.12.2019 14:31



Chemistry, 06.12.2019 14:31

Chemistry, 06.12.2019 14:31


English, 06.12.2019 14:31

Social Studies, 06.12.2019 14:31




Biology, 06.12.2019 14:31

History, 06.12.2019 14:31




