
Chemistry, 16.07.2020 03:01 adwinajames
The rate constant for this first‑order reaction is 0.550 s−10.550 s−1 at 400 ∘C.400 ∘C. A⟶products A⟶products How long, in seconds, would it take for the concentration of AA to decrease from 0.690 M0.690 M to 0.220 M?

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 22:30
Using the periodic table, complete the table to describe each atom. type in your answers.a ? b? c? d? e? f?
Answers: 1

Chemistry, 22.06.2019 18:00
Mercury turns to vapor at 629.88 k how much heat is lost 175 g of mercury vapor at 650 current condenses to a liquid at 297 ca mercury turns to weber at 629.88 kelvin how much he is lost 175 g of mercury vapor and 650 coming condensers to liquidate 297 kevin
Answers: 2

Chemistry, 22.06.2019 19:30
Estimate the molar mass of the gas that effuses at 1.6 times the effusion rate of carbon dioxide.
Answers: 1

Chemistry, 22.06.2019 19:30
What is the common name for the compound shown here? enter the common name of the compound shown?
Answers: 2
You know the right answer?
The rate constant for this first‑order reaction is 0.550 s−10.550 s−1 at 400 ∘C.400 ∘C. A⟶products A...
Questions



Mathematics, 06.03.2020 18:58


Mathematics, 06.03.2020 18:58





Computers and Technology, 06.03.2020 18:58




Mathematics, 06.03.2020 18:58






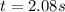
![ln(\frac{[A]}{[A]_0} )=kt](/tpl/images/0707/6088/00820.png)
![t=-\frac{ln(\frac{[A]}{[A]_0} )}{k}=- \frac{ln(\frac{0.220M}{0.690M} )}{0.55s^{-1}} \\\\t=-\frac{-1.14}{0.550s^{-1}}\\ \\t=2.08s](/tpl/images/0707/6088/3fd38.png)


