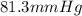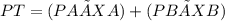Chemistry, 12.08.2020 06:01 danielahumajova6
At a given temperature the vapor pressures of benzene and toluene are 183 mm Hg and 59.2 mm Hg, respectively. Calculate the total vapor pressure over a solution of benzene and toluene with X Benzene

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 13:50
How does the motion of particles in a gas change as the gas cools
Answers: 2

Chemistry, 22.06.2019 21:00
What is the chemical formula for the compound formed between sodium and flour one
Answers: 1

Chemistry, 23.06.2019 00:00
How do you determine the percent yield of a chemical reaction
Answers: 1

Chemistry, 23.06.2019 01:00
Imagine if during the cathode ray experiment, the size of the particles of the ray was the same as the size of the atom forming the cathode. which other model or scientific observation would have also been supported? 1. this would support dalton's postulates that proposed the atoms are indivisible because no small particles are involved. 2. this would support bohr's prediction about electrons moving in orbits having specific energy. 3. this would support bohr's prediction about electrons being randomly scattered around the nucleus in the atom. 4. this would support dalton's postulates that proposed that atoms combine in fixed whole number ratios to form compounds.
Answers: 1
You know the right answer?
At a given temperature the vapor pressures of benzene and toluene are 183 mm Hg and 59.2 mm Hg, resp...
Questions

Mathematics, 16.10.2020 23:01

Computers and Technology, 16.10.2020 23:01


Mathematics, 16.10.2020 23:01


Mathematics, 16.10.2020 23:01


Mathematics, 16.10.2020 23:01


Biology, 16.10.2020 23:01

Mathematics, 16.10.2020 23:01

Computers and Technology, 16.10.2020 23:01

Medicine, 16.10.2020 23:01


Mathematics, 16.10.2020 23:01




Advanced Placement (AP), 16.10.2020 23:01